- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
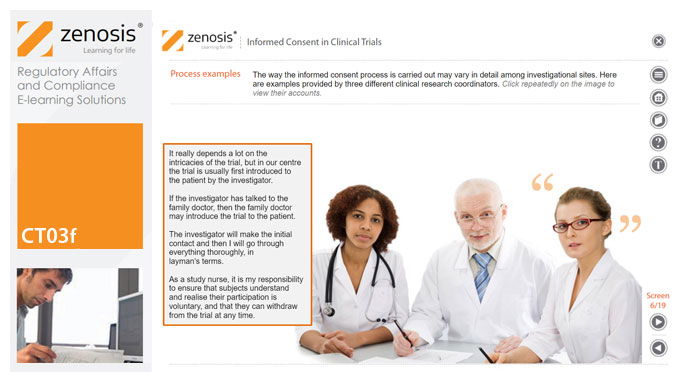
CT03f - Informed consent in clinical trials
By Zenosis
Informed consent in clinical research is an ethical and regulatory requirement. A research subject must enter a study voluntarily, be informed about risks and benefits, and understand the difference between investigation and treatment. Subjects must not be coerced into enrolment, nor must they be enticed by exaggerated claims of benefit. Before they can enrol, all potential subjects must agree, in writing, to participate. In addition to ethical and regulatory imperatives, the potential for litigation by subjects further highlights the importance of rigorous adherence to informed consent principles. In this short course we set out the principles and requirements and provide examples of practical issues confronting healthcare professionals and subjects.
Recruitment & HR Management at QLS level 3 & 5
By Compliance Central

Essentials in UX and HCD Management (CPUX-M)
By Bunnyfoot
This one-day course introduces the field of user experience and provides an excellent entry point to our other specialised training courses. UX processes and practices have become a central component of product design, service design and web design.

Level 3 Award in Education and Training: Elevate your career as a certified educator! Gain essential teaching skills, curriculum expertise, and effective classroom management. Flexible study options with ongoing support. Enrol now to advance your teaching journey

Journalism at QLS Level 6 Course
By One Education




