- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
4692 Courses
QUALIFI Level 2 Diploma in Business Beginners in Cyber Security
By School of Business and Technology London

QLS Endorsed Game Developer Bundle
By Imperial Academy
Game development is a symphony of code, graphics, and creativity, culminating in an epic journey for the player

React Bootcamp with .NET API (Router, Redux Toolkit, Hooks)
By Packt
This comprehensive course is designed to take you on a journey of mastering React, the popular JavaScript library for building dynamic user interfaces, and combining it with the power of .NET API for seamless back-end integration. This course will provide you with a solid foundation and hands-on experience in building full-stack applications.

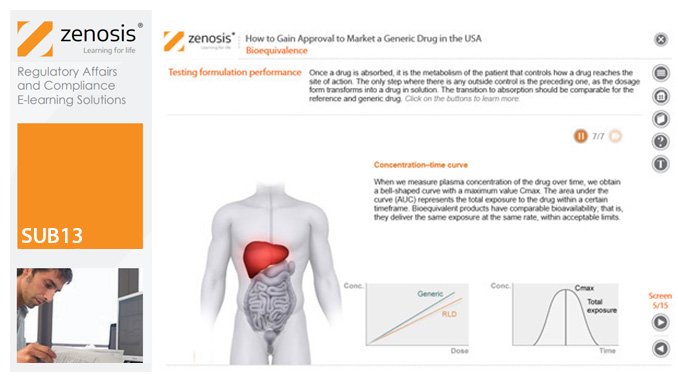
SUB13: How to Gain Approval to Market a Generic Drug in the USA
By Zenosis
This module outlines the legislative and regulatory context for the development of generic drugs and describes the essential role of the Abbreviated New Drug Application (ANDA) in gaining marketing approval. The use of information in the ‘Orange Book’ is explained, as is the role of patent certification in the application. The importance of establishing bioequivalence between a generic and its reference product is emphasised. The module specifies the content and format requirements for an ANDA submission and describes the FDA’s review and approval process. An outline is given of the Generic Drug User Fee Amendments (GDUFA) and the law’s effects on industry players.
Admin, Secretarial & PA (Executive PA) - 27 Courses with Free Certifications
By Training Express

Level 4 Diploma in Art Therapy - QLS Endorsed
By Kingston Open College
QLS Endorsed + CPD QS Accredited - Dual Certification | Instant Access | 24/7 Tutor Support | All-Inclusive Cost

UX Research
By Experience Thinkers
Arm yourself with a robust research toolkit that will help you uncover deep behavioural insights on user needs and motivations so you can design better experiences.

Journey Mapping
By Experience Thinkers
Arm yourself with a robust research toolkit that will help you uncover deep behavioural insights on user needs and motivations so you can design better experiences.

QUALIFI Level 3 Integrated Diploma in Business and Management
By School of Business and Technology London

Search By Location
- applications Courses in London
- applications Courses in Birmingham
- applications Courses in Glasgow
- applications Courses in Liverpool
- applications Courses in Bristol
- applications Courses in Manchester
- applications Courses in Sheffield
- applications Courses in Leeds
- applications Courses in Edinburgh
- applications Courses in Leicester
- applications Courses in Coventry
- applications Courses in Bradford
- applications Courses in Cardiff
- applications Courses in Belfast
- applications Courses in Nottingham
