- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
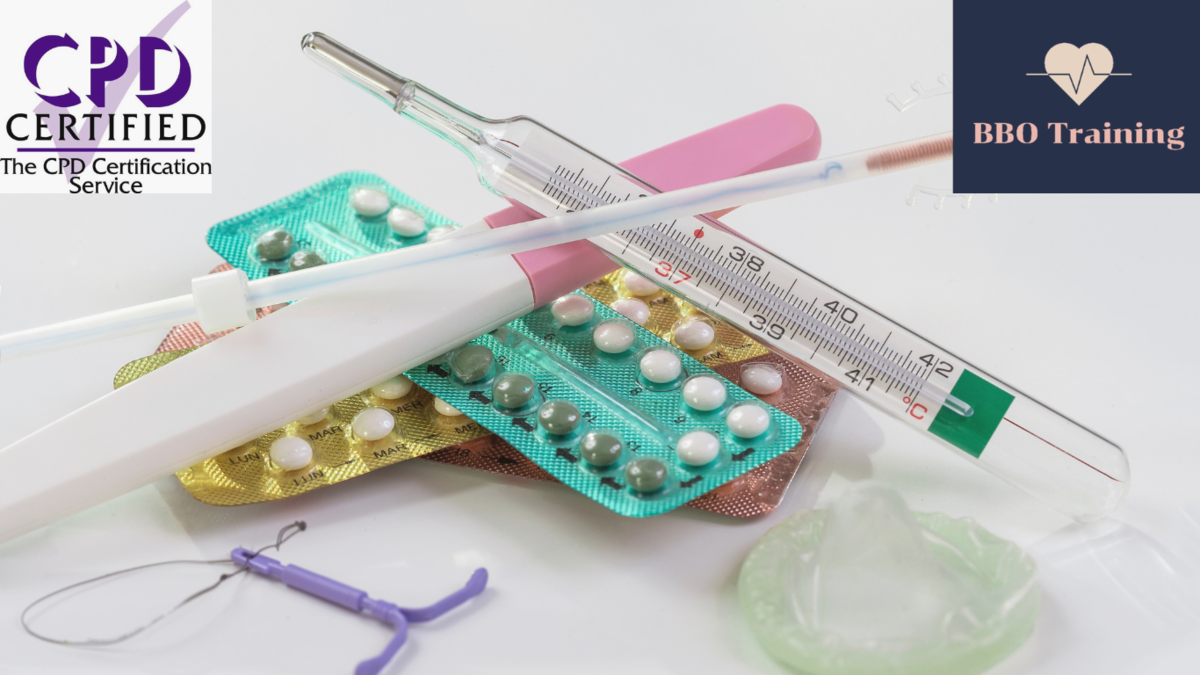
Introduction to Contraception for Healthcare Professionals (CPD Accredited)
By BBO Training
Level 5 Diploma in Education and Training (DET - Previously DTLLS) - Job Ready Program
4.8(12)By Academy for Health and Fitness

Vectorworks Landmark Training
By London Design Training Courses

SUB04: Preparing Submissions in the Common Technical Document (CTD) Format
By Zenosis
The CTD is the internationally recognised standard format for submissions to medicines regulatory authorities. In the European Economic Area, the USA and Canada, the CTD, in its electronic format (eCTD), is mandatory for all applications for marketing approval and all subsequent related submissions. The CTD is accepted in many other countries, being mandatory for new prescription medicines in some. This module explains the rationale for the CTD and provides guidance on its structure and format and the ways in which it is used.

Studio Health and Safety for Potters - managing dust, fumes, poisons, ...
By Tim Thornton
Studio Health and Safety for Potters - an online course by Tim Thornton. This covers the risks of dust, fumes, toxic materials and more, and how to deal with them

SEO 2020 - The Complete WordPress SEO Blueprint
By Packt
Let your online presence reflect your services, products, and articles by developing a successful online business or website using effective search engine optimization (SEO) strategies! This course will help you move your website and content to the first page of Google and other major search engines by getting you up to speed with the latest SEO techniques.

This full day workshop is designed to follow on and build on the learning from the 1-hour webinar to provide an embedded learning experience leading to acceptance and change of culture around neurodiversity. We understand the pressure managers can experience working within a neurodiverse team, this training is designed with managers in mind.

Infection Prevention, PUWER and Cleaning Diploma
By Imperial Academy
3 QLS Endorsed Diploma | QLS Hard Copy Certificate Included | 10 CPD Courses | Lifetime Access | 24/7 Tutor Support

Pipeline Integrity Management - Incorporating API 1160 / 1173 and ASME B31.8S
By EnergyEdge - Training for a Sustainable Energy Future

