542 Medicine & Nursing courses
Get a 10% discount on your first order when you use this promo code at checkout: MAY24BAN3X
Practical Approach to Auditing Systems and Processes
By Research Quality Association
COURSE INFORMATION Our extensively proven course delves into the essential stages of process and system auditing. Gain invaluable insights and direction in auditing systems and processes, spanning across global and local organisational levels. THIS COURSE WILL ASSIST DELEGATES WITH: * A practical approach for the development and conduct of process and system audits * An enhanced understanding of key system audit principles, preparation, design and conduct * Increased expertise, efficiency and confidence. THIS COURSE IS STRUCTURED TO ENCOURAGE DELEGATES TO: * Discuss and develop ideas * Solve specific problems * Share knowledge and experiences. BY THE END OF THE COURSE DELEGATES WILL BE BETTER ABLE TO: * Design and plan more effectively to achieve their process and systems audit objectives and add value to their organisation * Improve the effectiveness, focus and credibility of the audit programme * Understand the key system audit principles, preparation, design and conduct * Develop system audit tools to ensure more effective audit conduct and outcome * Create audit strategies utilising risk management principles * Prepare for inspections. -------------------------------------------------------------------------------- TUTORS Tutors will be comprised of (click the photos for biographies): ALLISON JACK Executive Director, Bristol Myers Squibb ROCIO CASTELLANOS Director, Pfizer Ltd GUY HOUBEN G(C)LP Auditor, Janssen Pharmaceutical Companies of Johnson & Johnson -------------------------------------------------------------------------------- PROGRAMME Please note timings may be subject to alteration. DAY 1 08:50 REGISTRATION 09:00 WELCOME AND INTRODUCTIONS, EXPECTATIONS/CHALLENGES/EXPERIENCES A discussion to explore the range of approaches to the conduct of systems audit. 09:30 INTRODUCING SYSTEMS AUDIT What is a system? Why conduct system audits? Advantages, disadvantages and challenges. 10:20 BREAK 10:35 SYSTEMS AUDIT DESIGN AND PLANNING Identifying the customer, setting objectives, development of the audit plan and audit tools, plans for the audit report. 12:00 DESIGNING SYSTEM AUDIT TOOLS 12:45 LUNCH 13:30 SYSTEM AUDIT PLAN - EXERCISE 14:00 INTRODUCTION TO CASE STUDIES The objectives of the case studies are defined and process and outputs described. 14:15 CASE STUDIES - SESSION 1 A first opportunity for work on case studies. Defining objectives and scope and understanding the requirements of the audit client. 15:00 BREAK 15:20 CASE STUDIES - SESSION 1 CONTINUED 16:30 CASE STUDIES - FEEDBACK 17:00 CLOSE OF DAY 1 DAY 2 09:00 SIMPLE SYSTEM AUDIT EXAMPLE - INTRODUCTION The objectives of the case studies are defined and process and outputs described. 09:10 CASE STUDIES - SESSION 2 - A SIMPLE SYSTEM AUDIT EXAMPLE An example of system audit applied to a simple system. 10:30 BREAK 10:45 A SIMPLE SYSTEM AUDIT EXAMPLE - CASE STUDY FEEDBACK 11:30 STRATEGY Audit programme planning. 12:15 LUNCH 13:00 CASE STUDIES - SESSION 3 Work on delegate's case studies. 14:30 BREAK 14:45 CASE STUDIES - SESSION 3 - FEEDBACK 15:15 CLOSING REMARKS 15:30 CLOSE OF COURSE -------------------------------------------------------------------------------- EXTRA INFORMATION Face-to-face course COURSE MATERIAL Course material will be available in PDF format for delegates attending this course. The advantages of this include: * Ability for delegates to keep material on a mobile device * Ability to review material at any time pre and post course * Environmental benefits – less paper being used per course. The material will be emailed in advance of the course and RQA will not be providing any printed copies of the course notes during the training itself. Delegates wishing to have a hard copy of the notes should print these in advance to bring with them. Alternatively delegates are welcome to bring along their own portable devices to view the material during the training sessions. Remote course COURSE MATERIAL This course will be run completely online. You will receive an email with a link to our online system, which will house your licensed course materials and access to the remote event. Please note this course will run in UK timezone. THE ADVANTAGES OF THIS INCLUDE: * Ability for delegates to keep material on a mobile device * Ability to review material at any time pre and post course * Environmental benefits – less paper being used per course Access to an online course group to enhance networking. You will need a stable internet connection, a microphone and a webcam. CPD POINTS 14 Points DEVELOPMENT LEVEL Develop

Good Laboratory Practice for Study Directors, Principal Investigators, Study Staff and Management
By Research Quality Association
COURSE INFORMATION Embark on our GLP course offering extensive guidance and pragmatic support tailored for individuals serving as Study Directors or Principal Investigators overseeing non-clinical safety studies on pharmaceuticals, agricultural, and industrial chemicals within the realm of Good Laboratory Practice (GLP). This comprehensive programme extends its benefits to study staff and management operating in GLP-compliant environments. The course extensively covers the current OECD GLP Principles and UK GLP legislation, while also referencing international standards, regulations, and guidelines pertinent to the field. BENEFITS OF THIS COURSE: * Practical help and guidance on the interpretation and application of GLP * An opportunity to update your knowledge of GLP with the current interpretation of requirements * Access to an experienced panel of speakers * Information on how other organisations address GLP issues * An opportunity to improve your understanding of the GLP requirements as they are applied in different situations. THIS COURSE IS STRUCTURED TO ENCOURAGE DELEGATES TO: * Discuss and develop ideas * Solve specific problems * Examine particular aspects of GLP * Learn from the experience of others. -------------------------------------------------------------------------------- TUTORS Tutors will be comprised of (click the photos for biographies): TIM STILES Consultant, Qualogy Ltd TONY WOODALL Head of Quality Assurance, Alderley Analytical GILL ARMOUR Study Monitor Team Leader, AstraZeneca JANE ELLISTON Senior Quality Assurance Auditor, Battelle UK VANESSA GRANT -, - JEANET LOGSTED CEO, Scantox -------------------------------------------------------------------------------- PROGRAMME Please note timings may be subject to alteration. DAY 1 09:00 REGISTRATION 09:15 WELCOME AND INTRODUCTIONS 09:35 DEVELOPMENT OF GOOD LABORATORY PRACTICE A review of the history of GLP, its current scope and application, with a synopsis of current European and international standards. 10:05 ROLES AND RESPONSIBILITIES The responsibilities of study director, test facility, management and study staff in the conduct of a GLP study. 10:45 BREAK 11:00 THE ROLES AND RESPONSIBILITIES OF THE STUDY DIRECTOR AND TEST FACILITY MANAGEMENT The role of the study director in the management and control of a study, as defined by GLP, and management's roles are explored. 11:45 MULTI-SITE STUDIES What is a multi-site study and when should such concepts be applied on a study. The role of the study director and principal investigator in the planning, conduct and reporting of multi-site study are explored. 12:30 STUDY PLAN (PROTOCOLS) GLP requirements for the preparation of a study plan, content, authorisation, amendments and deviations are discussed. 13:00 LUNCH 13:45 WORKSHOP 1 - THE STUDY PLAN Some practical problems with study plans and amendments explored. 14:45 WORKSHOP 1 - FEEDBACK 15:00 STANDARD OPERATING PROCEDURES The control, content and authorisation of SOPs and the principles behind the practice. 15:30 BREAK 15:45 WORKSHOP 2 - PRACTICAL STUDY CONDUCT PROBLEMS Dealing with practical problems encountered during the conduct of studies. 16:40 WORKSHOP 2 - FEEDBACK 17:15 CLOSE OF DAY DAY 2 09:00 QUESTIONS AND ANSWERS Discussion of issues raised by course delegates. 09:20 QUALITY ASSURANCE The interactions between QA, management, study director and principal Investigator are discussed as is QAs role when conducting a multi-site study. 10:00 THE FINAL REPORT The content of the final report and the role of those involved in its preparation and approval. Specific reporting requirements when conducting a multi-site study are also explained. 10:30 BREAK 10:45 WORKSHOP 3 - FINAL REPORT PROBLEMS Practical problems of report preparation including compliance statements. 11:30 WORKSHOP 3 - FEEDBACK 12:00 MANAGEMENT OF RAW DATA AND RECORDS A view on how records and materials are managed and archived in compliance with GLP. 12:45 LUNCH 13:30 WORKSHOP 4 - DATA AND SAMPLE MANAGEMENT ISSUES Dealing with data and sample management issues. 14:15 WORKSHOP 4 - FEEDBACK 14:45 REGULATORY INSPECTION Government monitoring for compliance with Good Laboratory Practice. 15:15 PANEL SESSION This panel session will address any outstanding issues raised by delegates. 15:45 CLOSE OF COURSE -------------------------------------------------------------------------------- EXTRA INFORMATION Face-to-face course COURSE MATERIAL Course material will be available in PDF format for delegates attending this course. The advantages of this include: * Ability for delegates to keep material on a mobile device * Ability to review material at any time pre and post course * Environmental benefits – less paper being used per course. The material will be emailed in advance of the course and RQA will not be providing any printed copies of the course notes during the training itself. Delegates wishing to have a hard copy of the notes should print these in advance to bring with them. Alternatively delegates are welcome to bring along their own portable devices to view the material during the training sessions. Remote course COURSE MATERIAL This course will be run completely online. You will receive an email with a link to our online system, which will house your licensed course materials and access to the remote event. Please note this course will run in UK timezone. THE ADVANTAGES OF THIS INCLUDE: * Ability for delegates to keep material on a mobile device * Ability to review material at any time pre and post course * Environmental benefits – less paper being used per course Access to an online course group to enhance networking. You will need a stable internet connection, a microphone and a webcam. CPD POINTS 14 Points DEVELOPMENT LEVEL Develop

Advanced / Competency Phlebotomy Training
By Lead Academy
This advanced phlebotomy course will allow you to receive practical demonstrations and hands-on training as a part of the practical training to support your role. At the beginning of the session, you will be required to observe the experienced trainers and trained phlebotomists. You will learn how to smoothly draw blood, minimize hazards, and manage the patient’s reaction through a practical demonstration on a mannequin. Finally, under close supervision, you will get the opportunity to collect blood samples from other fellow colleagues. -------------------------------------------------------------------------------- Course Highlights * Gain a Recommendation Letter from Your Supervisor * Accredited by the CPD Certification * Recognised and Valuable Certification * Face-to-Face Practical Training Sessions * Supervised by experienced phlebotomists -------------------------------------------------------------------------------- LOCATION & CLASS SCHEDULE Venue Location Nr. 1: Bank Studio, 23 Park Royal Road, NW10 7JH You can select your preferred date from any of the following months: * 14th, 22nd, 29th May 2024 * 5th, 12th, 18th June 2024 * 16th, 24th June & 2nd July 2024 * 6th, 14th, 21st July 2024 * 17th, 24th, 31st July 2024 * 6th, 11th, 18th August 2024 * 22nd, 28th August & 3rd September 2024 * 8th, 15th, 22nd September 2024 * 18th, 24th September & 1st October 2024 * 6th, 13th, 20th October 2024 * 17th, 24th, 31st October, 2024 * 5th, 10th, 17th November, 2024 * 21st, 27th November & 3rd December, 2024 * 7th, 12th, 18th December, 2024 Venue Location Nr. 2: 44 Blucher Street, Birmingham, B1 1QJ You can select your preferred date from any of the following months: * 16th, 23rd, 30th May, 2024 * 18th, 25th, 30th July 2024 * 17th, 24th and 30th September, 2024 * 15th, 21th, 28th November, 2024 Venue Location Nr. 3: Pure Offices, Kembrey Park, Swindon, SN2 8BW You can select your preferred date from any of the following months: * 22nd, 29th June and 6th July 2024 * 10th, 17th, 24th August, 2024 * 12th, 19th, 26th October 2024 * 30th November, 7th December and 14th December 2024 It can be tough to put your best foot forward in the job market with only online Phlebotomy training. Hence, our practical sessions will provide you with the required experience that your employers might want to see when you apply for the job. This advanced phlebotomy competency training is designed to give you an opportunity to practice, build confidence and gain valuable experience. Which will help your professional career when you will apply for the job. This extensive course covers the following credentials: * Practical demonstration * Labelling tubes * Blood-taking techniques * Code of practice * Safety protocols * Needle safety techniques * Processing of blood samples * Prioritising patients -------------------------------------------------------------------------------- WHO IS THIS ADVANCED PHLEBOTOMY TRAINING COURSE FOR? This advanced phlebotomy course is primarily aimed at: * Phlebotomists * Health Visitors * Physiotherapists * Occupational Therapists * Healthcare professionals * Nurses, midwives, pharmacists and doctors * Freshers looking to begin their career as a phlebotomist * Anyone looking to enhance their blood-sampling skills Whether you are a fresher looking to kickstart your career in the field of phlebotomy or practising healthcare looking to enhance your phlebotomy skills, this course will help you achieve your professional aspirations by all means. AIMS AND LEARNING OUTCOMES OF THIS ADVANCED PHLEBOTOMY TRAINING COURSE By the end of this advanced phlebotomy course, you will: * Know how to utilise proper safety precautions * Assess a phlebotomist’s personal and professional attributes * Accept the core roles and obligations of a phlebotomist * Understand the code of conduct required to be followed by a phlebotomist * Know how to draw blood using the right procedure * Establish the process to handle venipuncture-related issues * Understand how to handle and preserve blood samples safely ENTRY REQUIREMENTS * In order to attend the advanced phlebotomy course you need to complete our basic phlebotomy training course [https://lead-academy.cademy.co.uk/phlebotomy-training?occurrence=713e1556-934e-4302-afbd-b40a2d7a548d]. * However, it is open to students of all academic backgrounds. ASSESSMENT METHOD As part of the assessment, you will be required to execute a live blood draw and collection that will be closely supervised and marked by the experienced phlebotomist responsible for monitoring you. Upon successful demonstration of blood sampling, you will be awarded a certificate of competency that is accepted by thousands of professional bodies and government regulators here in the UK and around the world. -------------------------------------------------------------------------------- RECOGNISED ACCREDITATION This course is accredited by Continuing Professional Development (CPD). CPD UK is globally recognised by employers, professional organisations, and academic institutions, thus a certificate from CPD Certification Service creates value towards your professional goal and achievement. CPD certificates are accepted by thousands of professional bodies and government regulators here in the UK and around the world. Many organisations look for employees with CPD requirements, which means, that by doing this course, you would be a potential candidate in your respective field. -------------------------------------------------------------------------------- CERTIFICATE OF ACHIEVEMENT COMPETENCY CERTIFICATE AND CERTIFICATE OF ACHIEVEMENT FROM LEAD ACADEMY You will receive a competency certificate after successfully completing the Part 2 of the phlebotomy course, which serves as confirmation that you are qualified to function as a phlebotomist without the need for close supervision. Also, on successful completion of the phlebotomy course, you will be eligible to obtain a CPD accredited PDF and Hardcopy certificate of achievement as proof of your new skill. The certificate of achievement is an official credential that confirms that you successfully finished a course with Lead Academy.

CPD Accredited, Interactive Short Course * 2 or 3.5 hour hr sessions * What are the autistic spectrum disorder, autism and asperger's syndrome? * How can you best understand, and thus help, those affected? Course Contents of the shorter course: * What is ASD * Autism and Asperger's Syndrome * Understanding those on the Spectrum * How to approach and help those on the Autistic Spectrum * Great for teachers and TAs in schools, and for those working in care with older people with ASD BENEFITS OF THIS SHORT COURSE: * About 1% of the population is thought to be on the Autistic Spectrum * That is several children per nursery and primary school and a significant amount of young people in secondary schools * Many with Asperger's Syndrome will also go to university * The way they experience the world is very different to 'neuro-typical' people * Understanding their condition helps to support them and give them the best chance

Laparoscopy Modular Course
By CCMIG
Modular laparoscopy course with stepwise curriculum. At home laparoscopy trainer for the duration of the course with hands on sessions followed by final assessment away day over 5 months. Integrated online curriculum via the CCMIG website.
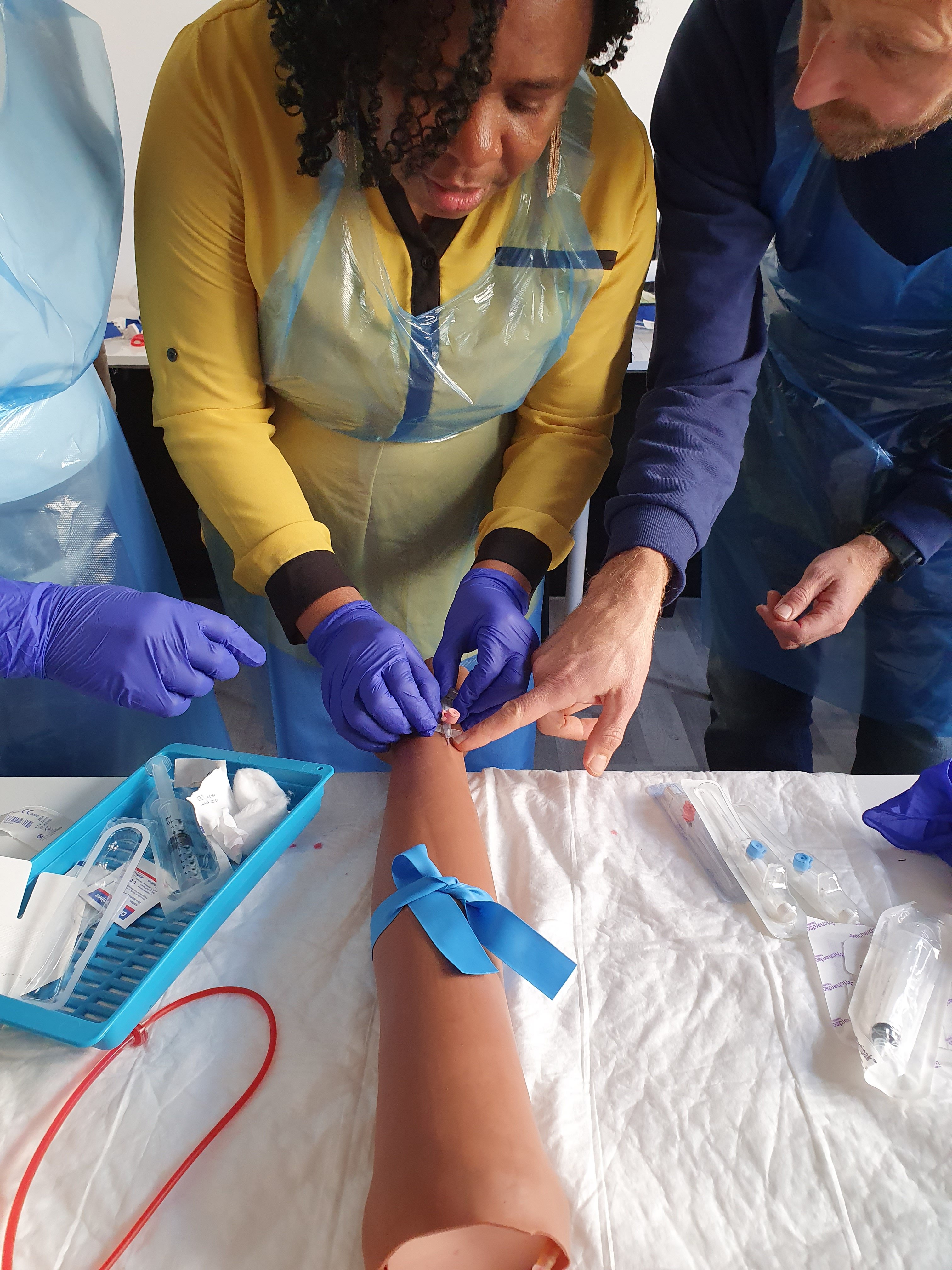
Venepuncture and cannulation course Venepuncture training for healthcare professionals Cannulation skills development Intravenous access techniques Blood sample collection training Infection control in venepuncture Hands-on venepuncture practice Cannula insertion training Nursing revalidation hours CPD accredited course Healthcare professional skills development Venepuncture and cannulation procedures Venepuncture certification program Intravenous catheter insertion Best practices in venepuncture Patient assessment for venepuncture Troubleshooting venepuncture complications Venepuncture and cannulation simulation Real-life venepuncture scenarios Healthcare career advancement with venepuncture skills
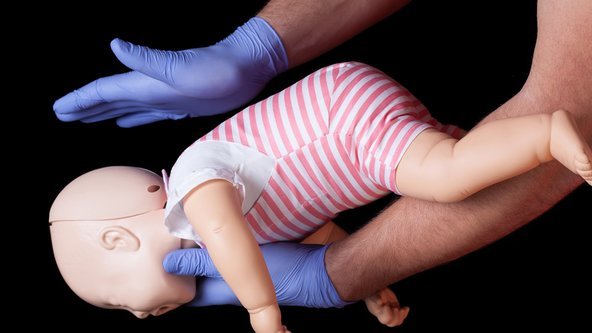
This course is ideal for anyone working in childcare – The Level 3 Award in Paediatric First Aid will give you the knowledge to act with confidence and how to deal with emergency situations in a safe and appropriate manner. This course covers the more common illnesses that occur in children as well as the types of incidents and accidents that could happen.
Health & Safety, PPE Covid COSHH & RIDDOR Equality & Diversity, Human Rights Infection Control Food Safety Information Governance Data Protection (GDPR) Records Management Fraud & Bribery Conflict Resolution Lone Worker Moving & Handling level 2(practical covered) Fire Safety Freedom of Information Safeguarding Adult & Children level 3 Radicalisation Slips Trips & Falls Your Healthcare Career Person Centred Care Duty of Care Consent Communication Privacy & Dignity Complaints Handling BLS –CPR & Choking for Adults and Children (practical covered) Mental Capacity Act—DoLS Mental Health Act

The UK's first and only Level 4 qualification in Phlebotomy (equivalent to Ireland Level 6) FDSc (Foundation Degree Level) qualification Nationally Recognised certificate Dually accredited: Open College Network and CPD Covers both aspirated and evacuated systems Covers specialised blood collection systems & methods Classroom or Virtual Classroom learning options Comprehensive Training Kit is provided when booking our Virtual Classroom option (yours to keep) Complete your training from beginner to advanced level This course either follows on from our Introduction to Phlebotomy Course or can be combined with our introductory course as part of a course package (see below) Available to candidates who have completed (or are currently enrolled to complete) our Introduction to Phlebotomy Course or have previous phlebotomy practical experience.
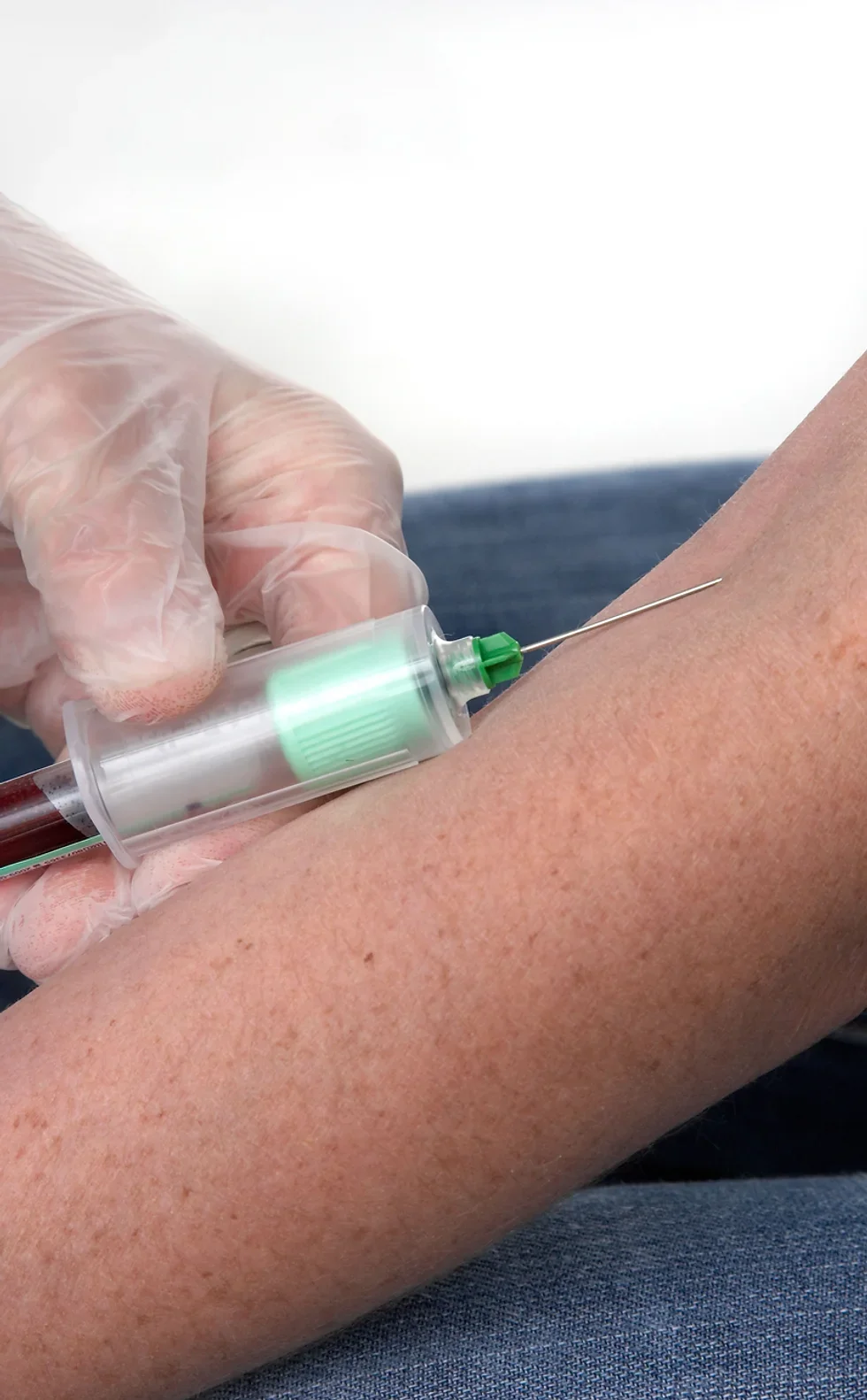
AVAILABLE ONLY TO APPLICANTS WHO HAVE ENROLLED ON (OR HAVE PREVIOUSLY COMPLETED) OUR INTRODUCTION TO PHLEBOTOMY COURSE Geopace Certificate of Competency Add live blood draws to your training Follow and complete a Competency Checklist Obtain tutor sign-off for live draws Awards a CPD Accredited Certificate Awards a Geopace Certificate of Competency Half day or evening sessions