- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
Courses in Nottingham
We couldn't find any listings for your search. Explore our online options below.
Know someone teaching this? Help them become an Educator on Cademy.
Online Options
Show all 43MD01: An Introduction to the Regulation of Medical Devices
By Zenosis
This module provides an introduction to the basics of medical device regulation, especially the requirements that manufacturers must meet in order to market devices in Europe and the USA.

ISO 13485 Quality Management Foundation
By Training Centre
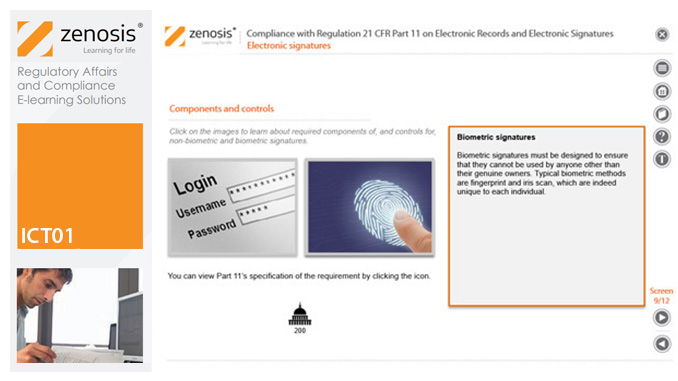
ICT01: Compliance with Regulation 21 CFR Part 11 on Electronic Records and Electronic Signatures
By Zenosis
21CFR11 applies to records that are required to be submitted to the FDA, or that are subject to FDA inspection, and that are in electronic form – that is, as computer files. It applies to all computer systems used to create, modify, maintain, archive, retrieve, or transmit such records – from a humble spreadsheet program to a complex information management system.
Medical Device Regulations (EU) Course
By One Education
Essentials EU Medical Device Regulations
By Course Gate
Explore the essentials of European Medical Device Regulations (EU MDR) with our comprehensive course. Gain insights into regulatory requirements, quality systems, and reporting protocols to ensure compliance in the healthcare industry.
Essentials of European Medical Device Regulations - CPD Certified Course
By One Education

Search By Location
- Medical devices Courses in London
- Medical devices Courses in Birmingham
- Medical devices Courses in Glasgow
- Medical devices Courses in Liverpool
- Medical devices Courses in Bristol
- Medical devices Courses in Manchester
- Medical devices Courses in Sheffield
- Medical devices Courses in Leeds
- Medical devices Courses in Edinburgh
- Medical devices Courses in Leicester
- Medical devices Courses in Coventry
- Medical devices Courses in Bradford
- Medical devices Courses in Cardiff
- Medical devices Courses in Belfast
- Medical devices Courses in Nottingham



