- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
Neurodiversity Workshop Developing: Learning, Development, Succession
By Mpi Learning - Professional Learning And Development Provider
A four-hour workshop on how to accelerate the learning and development of your Neurodiverse colleagues.

Delivered in Loughborough or UK Wide or OnlineLoughborough or UK WideorOnlineFlexible Dates
£445
HR Level 2 & 3 at QLS
By Imperial Academy
Level 3 QLS Endorsed Course | Endorsed Certificate Included | Plus 5 Career Guided Courses | CPD Accredited

Delivered Online On DemandOn-demand course
£129
Certified Kanban System Design – KMP I - 19-21 November
By Tom Reynolds
Attend our world class Kanban University accredited Certified Kanban System Design training course and learn to implement Kanban in your company

Delivered OnlineOnline courseTue, Nov 19 • 09:00
£1074 to £1194
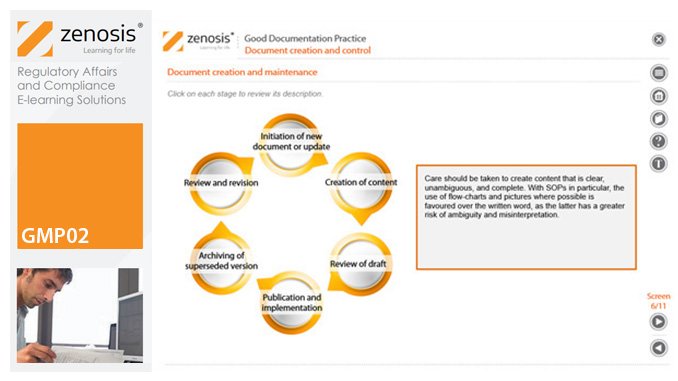
GMP02: Good Documentation Practice
By Zenosis
Good Manufacturing Practice (GMP) for medicinal products relies on documentation. Good Documentation Practice (GDocP) is that part of GMP that applies to the creation, maintenance, use, and retention of documents to provide assurance of the quality of products.
Delivered Online On DemandOn-demand course1 hour
£39
Fully Booked - How to Attract and Keep New Clients
By Dan Roberts

Delivered OnlineOnline masterclassFlexible Dates
£700
Project Management Foundation (PM Foundation): On-Demand
By IIL Europe Ltd

Delivered Online On DemandOn-demand course16 hours
£495
Neurodiversity Workshop Retaining: Reward, Change, Communication
By Mpi Learning - Professional Learning And Development Provider
A four-hour workshop on how to retain your talented Neurodiverse colleagues and ensure equality in employment opportunities.

Delivered in Loughborough or UK Wide or OnlineLoughborough or UK WideorOnlineFlexible Dates
£534


