- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
2324 Courses
OCC Access Course 2024
By Osteopathic Centre for Children

Delivered In-PersonIn-Person courseFlexible Dates
£200 to £600
Global Healthcare Management - Online Course
By One Education

Delivered Online On DemandOn-demand course3 days
£100
Professional Certificate Course in Sociowork in Communities Individuals and Families in London 2024
4.9(261)By Metropolitan School of Business & Management UK

Delivered Online On DemandOn-demand course14 days
£23
Lab Technician at QLS Level 7 Diploma
By Imperial Academy
Level 7 QLS Endorsed Diploma | 11-in-1 Bundle | CPD Accredited | Lifetime Access | Tutor Support

Delivered Online On DemandOn-demand course
£49
DNA and Genealogy Course
By Apex Learning

Delivered Online On DemandOn-demand course3 hours 6 minutes
£12
Hospital Receptionist & Secretary
By Training Tale

Delivered Online On DemandOn-demand course3 days
£49
PRP / PRF SKIN & HAIR COURSE
By Harley Elite Academy (HeLa)
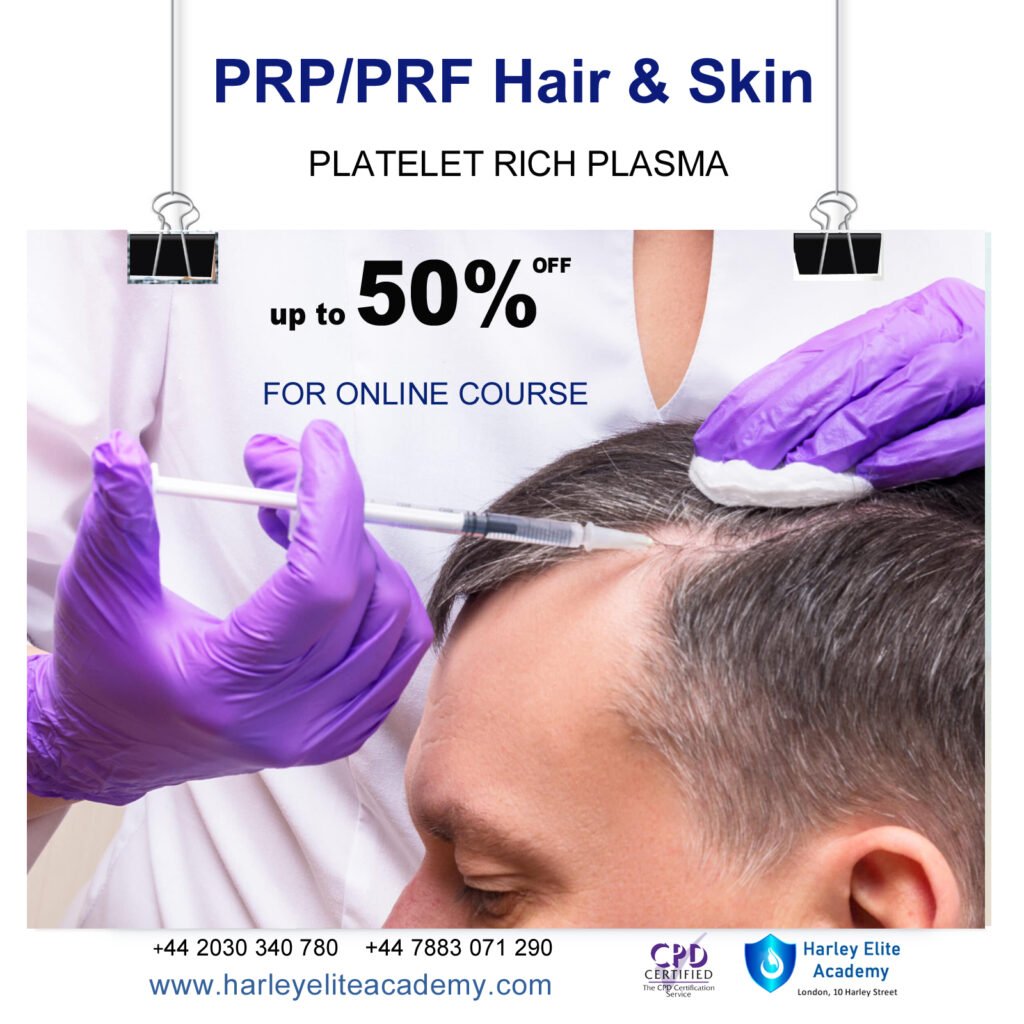
Delivered in London or UK Wide or OnlineLondon or UK WideorOnlineFlexible Dates
£700 to £1390
PV04: Signal Detection and Management in Pharmacovigilance
By Zenosis
This module provides a guide to signal detection and management for approved products. The subject is presented as a process comprising four stages: signal detection, signal validation, signal analysis and prioritisation, and risk assessment and minimisation.

Delivered Online On DemandOn-demand course1 hour 30 minutes
£74
ICT01: Compliance with Regulation 21 CFR Part 11 on Electronic Records and Electronic Signatures
By Zenosis
21CFR11 applies to records that are required to be submitted to the FDA, or that are subject to FDA inspection, and that are in electronic form – that is, as computer files. It applies to all computer systems used to create, modify, maintain, archive, retrieve, or transmit such records – from a humble spreadsheet program to a complex information management system.
Delivered Online On DemandOn-demand course1 hour 30 minutes
£74
Search By Location
- Clinical Courses in London
- Clinical Courses in Birmingham
- Clinical Courses in Glasgow
- Clinical Courses in Liverpool
- Clinical Courses in Bristol
- Clinical Courses in Manchester
- Clinical Courses in Sheffield
- Clinical Courses in Leeds
- Clinical Courses in Edinburgh
- Clinical Courses in Leicester
- Clinical Courses in Coventry
- Clinical Courses in Bradford
- Clinical Courses in Cardiff
- Clinical Courses in Belfast
- Clinical Courses in Nottingham
