- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
2919 Courses
CT03c - Clinical trial documentation
By Zenosis
Regulatory authorities tend to abide by the maxim that ‘If it isn’t documented, it didn’t happen’. Rigorous documentation of all aspects of a clinical trial is necessary to provide evidence of GCP and compliance with regulatory requirements, as well as enabling effective management of the trial. In this short course we describe important examples of the documents designated by ICH GCP as essential to the conduct of a clinical trial.

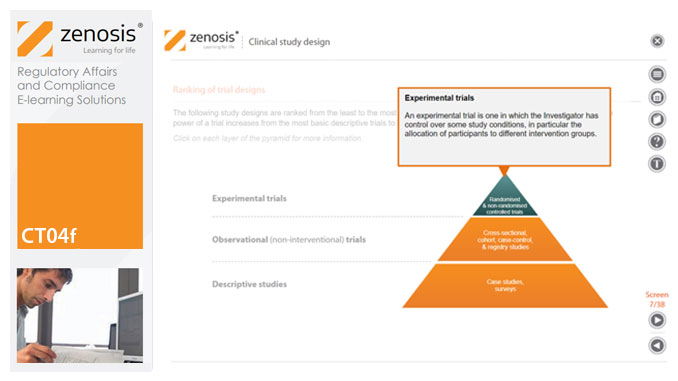
CT04f - Clinical study design
By Zenosis
Clinical trial design establishes the framework upon which the clinical trial process will be conducted, and sets the objectives of the trial. The application for marketing approval, submitted to the regulatory authorities, will provide clinical data reflecting the trial design. Since trial design impacts the whole drug development process and lifecycle, particular care and due diligence is essential. This short course provides an overview of the main types of study design.
Level 3, 4 & 5 Mental Health Care - MCA & DOLS
By Imperial Academy
Level 5 QLS Endorsed Course with FREE Certificate | CPD & CiQ Accredited | 150 CPD Points | Lifetime Access

Level 3, 4, 5 Mental Health Care - MCA & DOLS
By Imperial Academy
Level 5 QLS Endorsed Course with FREE Certificate | CPD & CiQ Accredited | 150 CPD Points | Lifetime Access

Level 2, 3 and 5 Mental Capacity Act
By Imperial Academy
Level 5 QLS Endorsed Course with FREE Certificate | CPD & CiQ Accredited | 150 CPD Points | Lifetime Access

The Complete Ethical Hacking Course
By Packt
If you are a newbie in the field of ethical hacking or want to become an ethical hacker, this course is just what will get you started. This is a comprehensive course with real-world examples to help you understand the fundamentals of hacking and cyber security.

How to make AI work for you - Chelmsford
By Let’s Do Business Group
Join us this November for a FREE in-person workshop in Chelmsford, which introduces SMEs to a variety of AI tools useful for improving business processes!

Social Media for Health & Care at QLS Level 4 Course
By One Education

Level 3 Certificate in Social Housing and Tenant Management
By Compliance Central

Search By Location
- author Courses in London
- author Courses in Birmingham
- author Courses in Glasgow
- author Courses in Liverpool
- author Courses in Bristol
- author Courses in Manchester
- author Courses in Sheffield
- author Courses in Leeds
- author Courses in Edinburgh
- author Courses in Leicester
- author Courses in Coventry
- author Courses in Bradford
- author Courses in Cardiff
- author Courses in Belfast
- author Courses in Nottingham
