- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
22565 Courses
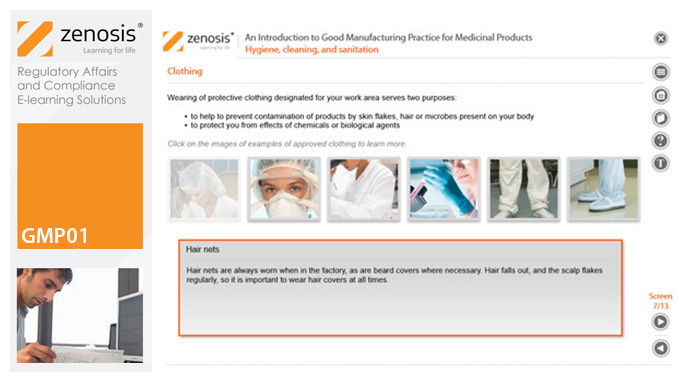
GMP01: An Introduction to Good Manufacturing Practice for Medicinal Products
By Zenosis
Good Manufacturing Practice (GMP) is a set of rules for medicines manufacturers to follow so that their products are safe, effective, and of good quality. The rules may be written into law or set out in guidance documents from regulatory authorities. Regulators will not allow medicinal products to be placed, or to remain, on the market in their country unless the products can be shown to be manufactured in compliance with GMP. To this end, they carry out inspections of manufacturing plants. Companies that persistently commit serious breaches of GMP requirements have suffered huge fines.
The Comprehensive Guide to Cabin Crew Careers - CPD Certified
4.8(12)By Academy for Health and Fitness

PKPD01: An Introduction to Pharmacokinetics and Pharmacodynamics in Drug Development and Registration
By Zenosis
Pharmacokinetic (PK) and pharmacodynamic (PD) studies provide a bridge between science and medicine in the development of a drug. In this module we describe the role of in-vivo PK and PD studies in a drug development programme, set out the uses to which the findings can be put, and discuss their implications for clinical development and application for marketing approval.

Selenium WebDriver 4 with Python - Zero To Hero
By Packt
This course will help you get started with automation testing of web applications. You will cover the basic and advanced topics of Selenium and Python, along with unit tests, pytest, cross-browser testing, logging infrastructure, automation framework design, Jenkins, and a lot more.

C204- WEATHERTIGHTESS AND TESTING (1/2 DAY)
By CWCT

Software Security Testing
By Packt
Learn how to perform security audits, penetration testing, and code auditing for modern software in the web/cloud, mobile, and desktop. This course will help you master industry standards and enable you to carry out professional assessments to secure technologies as well as communicate risks to high-level executives, management, and technical staff.

Search By Location
- Introduction to Plant Science: Essential Concepts Courses in London
- Introduction to Plant Science: Essential Concepts Courses in Birmingham
- Introduction to Plant Science: Essential Concepts Courses in Glasgow
- Introduction to Plant Science: Essential Concepts Courses in Liverpool
- Introduction to Plant Science: Essential Concepts Courses in Bristol
- Introduction to Plant Science: Essential Concepts Courses in Manchester
- Introduction to Plant Science: Essential Concepts Courses in Sheffield
- Introduction to Plant Science: Essential Concepts Courses in Leeds
- Introduction to Plant Science: Essential Concepts Courses in Edinburgh
- Introduction to Plant Science: Essential Concepts Courses in Leicester
- Introduction to Plant Science: Essential Concepts Courses in Coventry
- Introduction to Plant Science: Essential Concepts Courses in Bradford
- Introduction to Plant Science: Essential Concepts Courses in Cardiff
- Introduction to Plant Science: Essential Concepts Courses in Belfast
- Introduction to Plant Science: Essential Concepts Courses in Nottingham



