- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
Computer System Validation Training Course (ONLINE). Extend Your Role to CSV Projects. Get Certified and Become a CSV Professional
By Getreskilled (UK)
Has the Computer System Validation Engineer left and you’ve been handed their responsibilities? Do the thoughts of your next audit fill you with dread? CSV can be frustrating but this program will show you how to manage electronic data in a regulated manufacturing/laboratory/clinical environment using the GAMP framework and ensure compliance with FDA’s 21 CFR Part 11, EU Annex 11 or other regulatory guidelines.

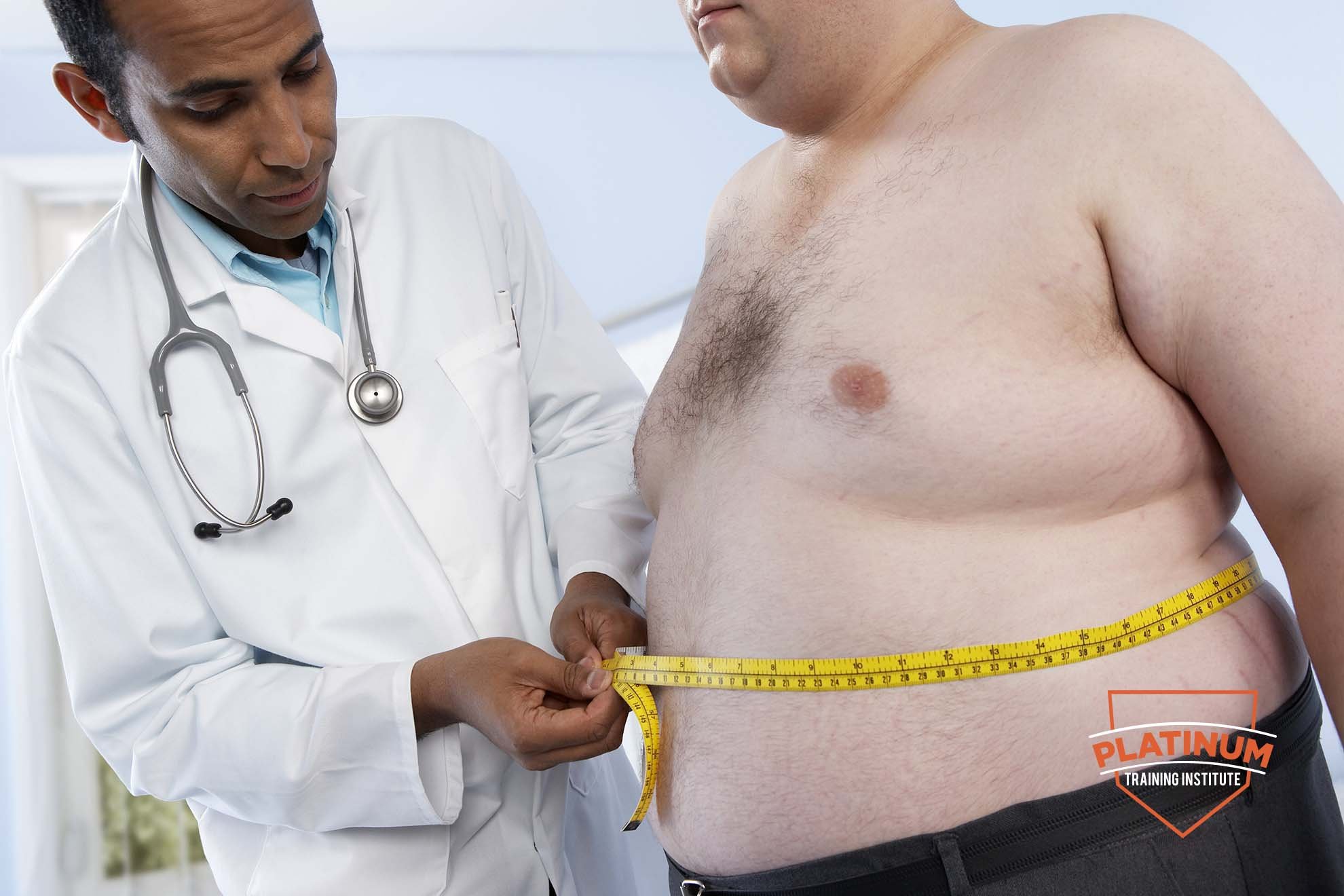
YMCA Level 4 Certificate in Weight Management for Individuals with Obesity, Diabetes Mellitus and/or Metabolic Syndrome
5.0(7)By Platinum Training Institute
This qualification is for those who wish to be able to plan and deliver safe and effective weight management programmes to members of the population with Obesity, Diabetes Mellitus and/or Metabolic Syndrome.
Mental Health Skills For Managers
By Starling
Learn how to create a supportive and open environment for mental health and wellbeing at work, helping everyone thrive and perform at their best.

AgileSHIFT: In-House Training
By IIL Europe Ltd

AET Level 3 Award in Education and Training
By Training Tale

APM Project Management Qualification (PMQ): Virtual In-House Training
By IIL Europe Ltd

Game Design Training: 3ds Max and Unity 3D Personalized
By Real Animation Works
Game design training face to face training customised and bespoke.

SAFe for Architects: Virtual In-House Training
By IIL Europe Ltd


