- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
Functional Skills Maths Training
By iStudy UK

UX (User Experience) Fundamentals Course
By NextGen Learning

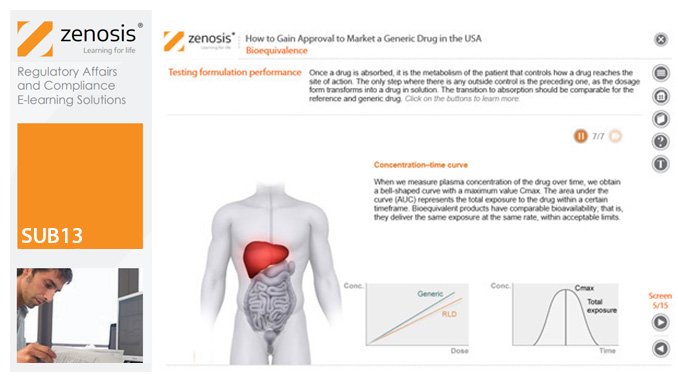
SUB13: How to Gain Approval to Market a Generic Drug in the USA
By Zenosis
This module outlines the legislative and regulatory context for the development of generic drugs and describes the essential role of the Abbreviated New Drug Application (ANDA) in gaining marketing approval. The use of information in the ‘Orange Book’ is explained, as is the role of patent certification in the application. The importance of establishing bioequivalence between a generic and its reference product is emphasised. The module specifies the content and format requirements for an ANDA submission and describes the FDA’s review and approval process. An outline is given of the Generic Drug User Fee Amendments (GDUFA) and the law’s effects on industry players.
The Complete Python and PostgreSQL Developer Course
By Packt
Build 9 projects to master 2 essential and modern technologies: Python and PostgreSQL

SCRUM Master Certified Course (SMC®) Online
By Aspire People and Business Development
Scrum Developer Certified (SDC®) is an entry level certification for Scrum Team members (developers) and those who interact with any Scrum Team. The objective of this certification is to ensure that Scrum Team members know enough about Scrum to effectively contribute to a Scrum project. The courseware and preparation tools required for the certification exam will be available online on SCRUMstudy.com. An online proctored exam will be conducted at the end of the course.

Explore the rich tapestry of political ideologies in the course 'Political Ideas: Different Forms of Politics.' From liberalism to ecologism, delve into the core principles, historical contexts, and contemporary relevance of various political ideologies, fostering critical analysis and interdisciplinary perspectives.




