- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
1570 Courses delivered Online
Nursing Assistant & Nurse Prescribing Diploma (Online) - CPD Certified
4.8(12)By Academy for Health and Fitness

Delivered Online On DemandOn-demand course21 hours
£35
Level 2 - Safe Handling of Medication in Health & Social Care
By Training Tale

Delivered Online On DemandOn-demand course40 hours
£12
Efficient Freight Broker Training Course
By Training Express

Delivered Online On DemandOn-demand course
£12
Understanding the Safe Handling of Medication in Health & Social Care
By Training Tale

Delivered Online On DemandOn-demand course19 hours
£12
Level 3 Health and Social Care with Mental Care Training
By Kingston Open College
Premium Bundle of all Time | Ofqual Regulation + ATHE Awards + CPD Accreditation | Assessment & Tutor Support Included

Delivered Online On DemandOn-demand course3 weeks
£999
Care Management - Health and Social Care with Care Certificate Standards Course
By One Education

Delivered Online On DemandOn-demand course3 days
£109
Understand Complaints and FCA Regulations
By Study Academy

Delivered Online On DemandOn-demand course1 hour
£30
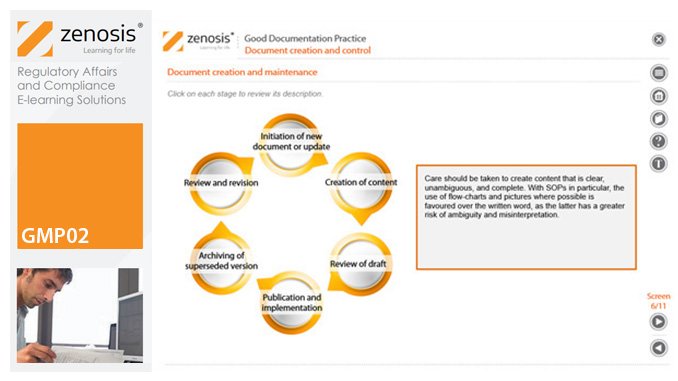
GMP02: Good Documentation Practice
By Zenosis
Good Manufacturing Practice (GMP) for medicinal products relies on documentation. Good Documentation Practice (GDocP) is that part of GMP that applies to the creation, maintenance, use, and retention of documents to provide assurance of the quality of products.
Delivered Online On DemandOn-demand course1 hour
£39

