- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
Explore the captivating world of Medieval literature with our course "Medieval Literature: A Journey with Chaucer and More." Dive into epic poems, romance narratives, and allegorical tales from renowned authors like Geoffrey Chaucer, Beowulf, and Dante Alighieri. Uncover themes of chivalry, courtly love, and religious symbolism while gaining insight into the cultural and historical contexts of the Middle Ages. Join us on this enriching journey through timeless masterpieces and expand your literary horizons.

Medical Transcription Diploma - CPD Certified
By Apex Learning

Digital Marketing: Affiliate Marketing, Social Media Marketing & SEO - (20 in 1) Course
By Compliance Central

ISO 29993:2017 Lead Auditor Training Course
By TUVSW Academy
ISO 29993:2017 Lead Auditor Training Course

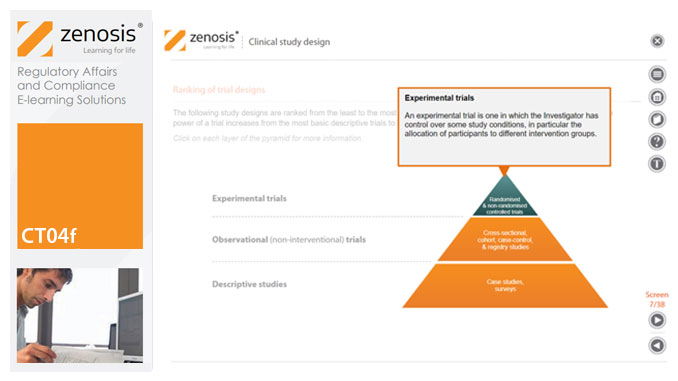
CT04f - Clinical study design
By Zenosis
Clinical trial design establishes the framework upon which the clinical trial process will be conducted, and sets the objectives of the trial. The application for marketing approval, submitted to the regulatory authorities, will provide clinical data reflecting the trial design. Since trial design impacts the whole drug development process and lifecycle, particular care and due diligence is essential. This short course provides an overview of the main types of study design.
CT03c - Clinical trial documentation
By Zenosis
Regulatory authorities tend to abide by the maxim that ‘If it isn’t documented, it didn’t happen’. Rigorous documentation of all aspects of a clinical trial is necessary to provide evidence of GCP and compliance with regulatory requirements, as well as enabling effective management of the trial. In this short course we describe important examples of the documents designated by ICH GCP as essential to the conduct of a clinical trial.

Level 3, 4 & 5 Mental Health Care - MCA & DOLS
By Imperial Academy
Level 5 QLS Endorsed Course with FREE Certificate | CPD & CiQ Accredited | 150 CPD Points | Lifetime Access

Level 3, 4, 5 Mental Health Care - MCA & DOLS
By Imperial Academy
Level 5 QLS Endorsed Course with FREE Certificate | CPD & CiQ Accredited | 150 CPD Points | Lifetime Access

Level 2, 3 and 5 Mental Capacity Act
By Imperial Academy
Level 5 QLS Endorsed Course with FREE Certificate | CPD & CiQ Accredited | 150 CPD Points | Lifetime Access

