- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
6743 Courses delivered Online
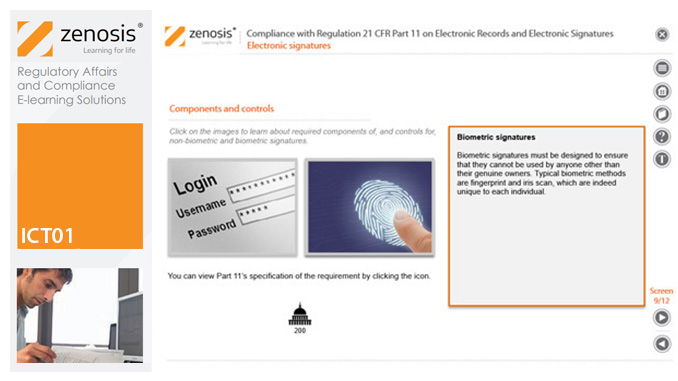
ICT01: Compliance with Regulation 21 CFR Part 11 on Electronic Records and Electronic Signatures
By Zenosis
21CFR11 applies to records that are required to be submitted to the FDA, or that are subject to FDA inspection, and that are in electronic form – that is, as computer files. It applies to all computer systems used to create, modify, maintain, archive, retrieve, or transmit such records – from a humble spreadsheet program to a complex information management system.
Close Protection, Security Guard, and Security Management - For 5 Employees
By Compliance Central

Diploma in Health and Safety - QLS Endorsed Course
By Training Express

PAT Testing - Portable Appliance Testing Training
By Course Gate
Explore the lucrative career of a PAT tester by enhancing your skills with our PAT Testing - Portable Appliance Testing Training course. Gain valuable insights into risk reduction and control, conduct visual inspections, and master the testing of PAT equipment.

Master the art of Credit Control with our comprehensive course! Explore strategies for Financial Stability, Credit Management, Risk Assessment, and cutting-edge Technologies. Enhance your skills in Debt Management, Customer Communication, Compliance, and Financial Analysis. Join us to gain expertise and contribute to your organization's success in today's dynamic financial landscape.





