- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
6747 Compliance courses delivered Online
Day Trading, Forex Trading and AML Course
By One Education

Day Trading, Forex Trading and AML Course
By One Education

Introducing GDPR-CPD Approved
By BAB Business Group
The General Data Protection Regulation (GDPR) is designed to strengthen and unify the principles of data protection for all individuals within the European Union and the European Economic Area. The GDPR is an incredibly important change to data privacy regulations so understanding its correct implementation is vital for all UK businesses and organisations, and particularly for staff who regularly deal with personal data. This online course is designed specially for those front line staff and provides a clear introduction to the main elements of the GDPR, including compliance and the consequences of non-compliance. It explains the roles of key players - Data Protection Officers, Data Controllers, Data Protection Leads and Data Processors and covers the main categories of personal data, along with the six lawful bases for processing data, and how to audit the data your organisation holds. Other topics examined include the Seven Principles of the GDPR and the Eight Rights for Individuals, along with the importance of your Privacy Policy - how to construct one, and how to use it effectively when dealing with data subjects. Finally, there's important information on data breaches; how to avoid them, what to do if one is discovered and how to file a breach report

Introduction to UK Financial Regulation and Professional Integrity
4.8(12)By Academy for Health and Fitness

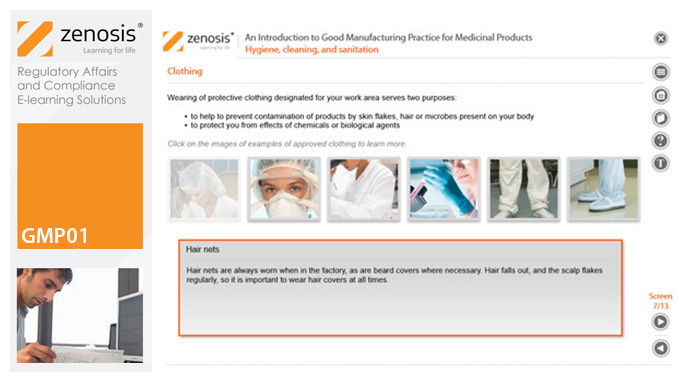
GMP01: An Introduction to Good Manufacturing Practice for Medicinal Products
By Zenosis
Good Manufacturing Practice (GMP) is a set of rules for medicines manufacturers to follow so that their products are safe, effective, and of good quality. The rules may be written into law or set out in guidance documents from regulatory authorities. Regulators will not allow medicinal products to be placed, or to remain, on the market in their country unless the products can be shown to be manufactured in compliance with GMP. To this end, they carry out inspections of manufacturing plants. Companies that persistently commit serious breaches of GMP requirements have suffered huge fines.
Chief Executive Officer (CEO) Career Track Diploma Course
By Training Express




