- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
4342 Courses
GTi Definition Brows
By The Beauty Click Academy

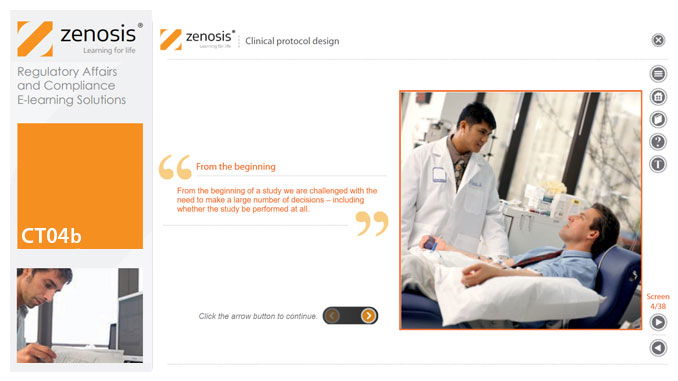
CT04b - Clinical protocol design
By Zenosis
Clinical trial protocols are an essential part of clinical trial design. Protocol documents are critical to conducting safe and cost-effective investigations. Protocol documents are large and complex, containing comprehensive information relating to purpose, design and conduct of a clinical trial. Aspects of a protocol include patient eligibility criteria, and treatment specifications. This short course provides an overview of clinical trial protocols. Opportunities to improve a clinical trial protocol for regulatory approval are also discussed.
RCN Accredited Asthma and COPD Course NORFOLK ICB ONLY
By BBO Training
An emergency can happen anywhere, so it's better to be prepared at all instances, specially at the workplace. Come to Knight Training and ensure your employees are safe with our Emergency First Aid At Work Course now!

Online Face & Body Waxing Training Course
By Cosmetic College

Level 3 Health and Social Care With Mental Health Awareness Diploma
By Kingston Open College
Premium Bundle of all Time | Ofqual Regulation + ATHE Awards + QLS Endorsement | Assessment & Tutor Support Included

Classic Swedish Massage- Diploma course
By Massage & Spa School
Search By Location
- Treatment Courses in London
- Treatment Courses in Birmingham
- Treatment Courses in Glasgow
- Treatment Courses in Liverpool
- Treatment Courses in Bristol
- Treatment Courses in Manchester
- Treatment Courses in Sheffield
- Treatment Courses in Leeds
- Treatment Courses in Edinburgh
- Treatment Courses in Leicester
- Treatment Courses in Coventry
- Treatment Courses in Bradford
- Treatment Courses in Cardiff
- Treatment Courses in Belfast
- Treatment Courses in Nottingham


