- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
2825 Courses
Work with Rachel to enhance your well-being and improve your quality of life. Reduce anxiety and stress, develop time management strategies, set goals, explore your purpose and values, improve confidence and self-worth, work on self-care and self-compassion, explore your limiting beliefs, regulate your emotions, manage chronic fatigue, improve your resilience, and much more.

Level 3 Award in Supporting Teaching and Learning with School Counselling Training
By Kingston Open College
Premium Bundle of all Time | Ofqual Regulation + NCFE Awards + QLS Endorsement | Assessment & Tutor Support Included

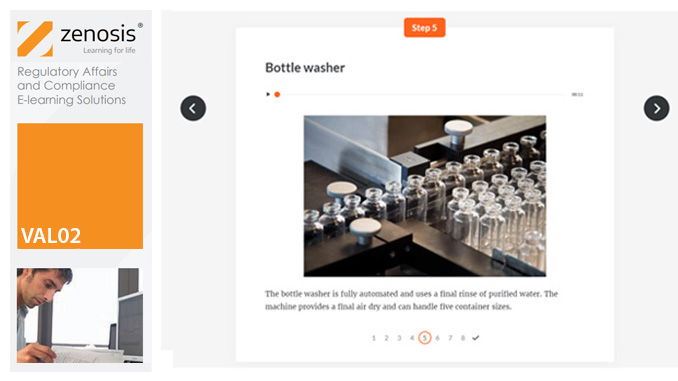
VAL02: Validation Plans and Documentation
By Zenosis
Essential to validation is the provision of documented evidence verifying that manufacturing processes will consistently result in products meeting predetermined quality standards. This module describes the purpose, content and use of validation master plans, project validation plans, and other documentation for validation projects in the medicines and healthcare products industries. It describes the activities of a typical validation team as they carry out a project for a pharmaceutical company.
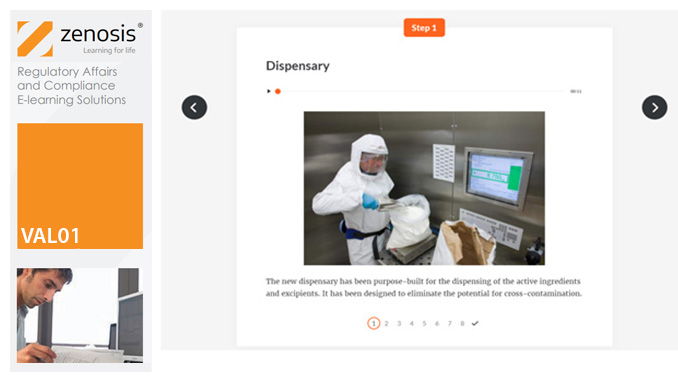
VAL01: Introduction to Validation
By Zenosis
Validation of equipment, services, systems and processes is vitally important in the medicines and healthcare products industries. Regulatory authorities require documented evidence that manufacturing processes will consistently result in products meeting predetermined quality standards. This module provides an introduction to validation and to the regulations and guidance that apply to it. It describes the activities of a typical validation team as they carry out a project for a pharmaceutical company.
Level 3 Award in Education and Training (RQF) (Former PTLLS) & SEN Course
By Apex Learning

Level 3 Award in Education and Training - AET (Formerly PTTLS)
By Apex Learning

Level 3 Award in Education and Training (RQF) - Train the Trainer (PTLLS)
By Apex Learning

Level 3 Award in Education & Training (RQF) and Special Educational Needs (SEN)
By Apex Learning

Search By Location
- EVI Courses in London
- EVI Courses in Birmingham
- EVI Courses in Glasgow
- EVI Courses in Liverpool
- EVI Courses in Bristol
- EVI Courses in Manchester
- EVI Courses in Sheffield
- EVI Courses in Leeds
- EVI Courses in Edinburgh
- EVI Courses in Leicester
- EVI Courses in Coventry
- EVI Courses in Bradford
- EVI Courses in Cardiff
- EVI Courses in Belfast
- EVI Courses in Nottingham