- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
39563 Courses
Social Work Form F Assessment Training CPD Accredited
By Sirius Group Training
Assessing potential foster carers can be a very enjoyable and rewarding role. Our CPD Accredited training course equips delegates with the skills needed to undertake a Form F assessment

New for 2020, this fun workshop will aim to move you on from “in-the-ditch” quilting and from quilting straight lines. Using a variety of techniques we will work on quilting common patchwork shapes to find a style and method that suits you best. This workshop is aimed at those with some quilting experience who are keen to move their skills on in a completely new direction. However, it is suitable for all abilities as full tuition and hand-holding will be provided on the day.

Cosmic Christ Series
By Katarina Heuser
Discover your special powers, receive insights and deep healing together with an abundance of light codes taking your life to the next level!

Level 3 Award in Understanding Assessment: Explore the core principles and practices essential for effective assessment. Enhance your knowledge of assessment methodologies and strategies. Flexible learning with comprehensive support. Enrol now to deepen your understanding! #AssessmentPrinciples #AssessmentPractices #EducationQualification

The Simplest Guide™ to Advanced SAS Programming | Macros | Proc SQL
By Packt
The Simplest Guide⢠to Advanced SAS Programming | Macros | SQL is an accelerated path to learning advanced SAS programming taught in a simple, visual, and immersive style. If you already know SAS, this course will equip you with skills to reveal amazing insights within your data, while helping you prepare for advanced SAS certification exams.

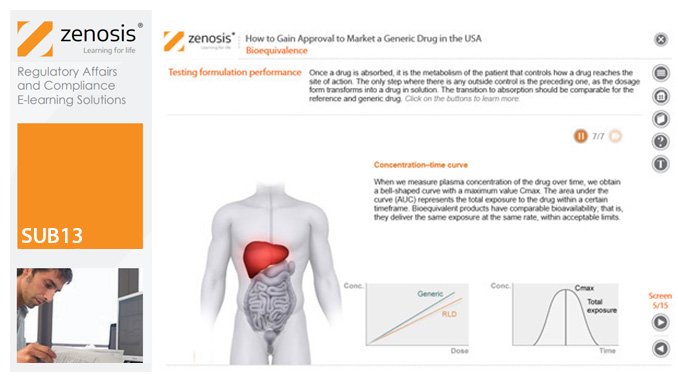
SUB13: How to Gain Approval to Market a Generic Drug in the USA
By Zenosis
This module outlines the legislative and regulatory context for the development of generic drugs and describes the essential role of the Abbreviated New Drug Application (ANDA) in gaining marketing approval. The use of information in the ‘Orange Book’ is explained, as is the role of patent certification in the application. The importance of establishing bioequivalence between a generic and its reference product is emphasised. The module specifies the content and format requirements for an ANDA submission and describes the FDA’s review and approval process. An outline is given of the Generic Drug User Fee Amendments (GDUFA) and the law’s effects on industry players.
Search By Location
- PAT Courses in London
- PAT Courses in Birmingham
- PAT Courses in Glasgow
- PAT Courses in Liverpool
- PAT Courses in Bristol
- PAT Courses in Manchester
- PAT Courses in Sheffield
- PAT Courses in Leeds
- PAT Courses in Edinburgh
- PAT Courses in Leicester
- PAT Courses in Coventry
- PAT Courses in Bradford
- PAT Courses in Cardiff
- PAT Courses in Belfast
- PAT Courses in Nottingham



