- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
2324 Courses
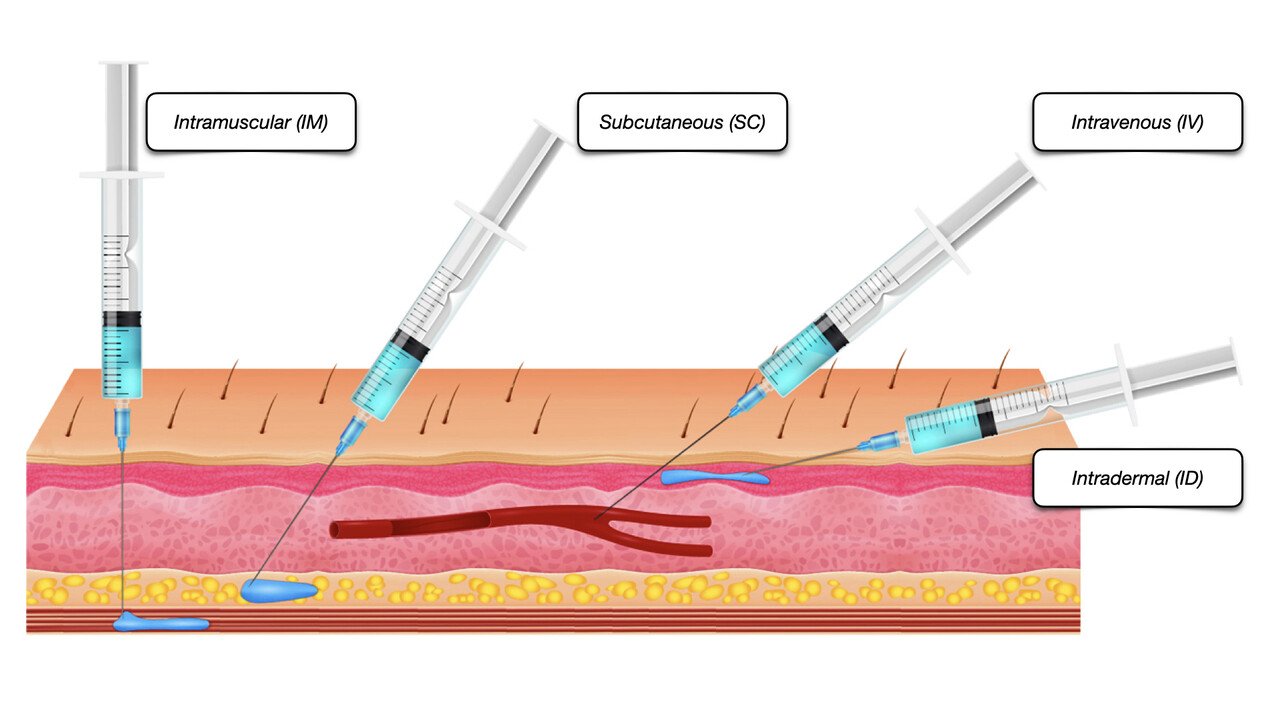
An Understanding of Injectable LHRH agonists
By Guardian Angels Training
Gain comprehensive knowledge on injectable LHRH agonists with our course. Learn about mechanisms, clinical applications, administration techniques, and more.
Clinical Psychology, Treatment and Therapy Course
By Training Express

Clinical Coding, Medical Coding, Medical Terminology, Anatomy and Physiology Training Course
By One Education

Clinical Nurse Training - Tissue Viability
By Course Gate
Gain expertise in tissue viability nursing with Clinical Nurse Training - Tissue Viability. Learn wound management, dressing selection, and pressure ulcer prevention strategies. Ideal for nurses seeking to enhance their skills and deliver exceptional patient care.

Module 5 part 1 Psychology & Neuroscience Workshop
5.0(19)By The Northern College Of Clinical Hypnotherapy
From exploring basic psychological phenomena like perception, memory, cognition, language, consciousness, and emotion to dissecting the latest advancements in brain imaging technology, this course offers a rapid deep dive into the intricacies of the human psyche. Gain a scientific understanding of the mind and behaviour while honing critical thinking skills essential for psychological inquiry.

CT10: The Investigational New Drug Application (IND) to Conduct FDA-regulated Clinical Trials
By Zenosis
An Investigational New Drug Application (IND) is a submission to the US Food and Drug Administration (FDA) for permission to conduct a clinical trial of a medicinal product. This module describes regulatory requirements that sponsors or sponsor-investigators must meet for successful compilation, filing and maintenance of INDs. The IND and its role are defined, and the contexts in which it is required are specified.
Search By Location
- Clinical Courses in London
- Clinical Courses in Birmingham
- Clinical Courses in Glasgow
- Clinical Courses in Liverpool
- Clinical Courses in Bristol
- Clinical Courses in Manchester
- Clinical Courses in Sheffield
- Clinical Courses in Leeds
- Clinical Courses in Edinburgh
- Clinical Courses in Leicester
- Clinical Courses in Coventry
- Clinical Courses in Bradford
- Clinical Courses in Cardiff
- Clinical Courses in Belfast
- Clinical Courses in Nottingham


