- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
23643 Courses
Essentials in Communication and Co-production with Parents/Carers (Request a date)
5.0(1)By Born at the Right Time
Testimonials “Powerful and inspirational” “Everyone would benefit from this training” "I will change how I work from this moment on." "I found your training extremely powerful and it has really impacted." Finalist for Best Educational Programme for the NHS HSJ Partnership Awards 2023 Essentials in Communication and Co-production with Parents/Carers is an inspiring, interactive and unique 4-hour virtual (or 6/7 hour in-person), CPD-certified training founded on lived experience, research and best practice. It aims to equip practitioners to effectively communicate and work in collaboration with parent/carers of children/adults who have disabilities. When time and resources are limited, maximising interactions with family/carers is essential in an age where co-production, personalised care and understanding lived experience are at the heart of delivering good services. This Born at the Right Time training course is hosted by Director/Founder Rachel Wright and aimed at anyone working in health, SEND or social care, whether a registered or non-registered practitioner. Content Current context for families and practitioners What is going on underneath the surface Next steps to creating meaningful change Trainer Rachel Wright (BSc RN) is founder of Born at the Right Time, author of The Skies I'm Under, award winning blogger and Best Trainer finalist 2022 National Learning Disability and Autism Awards. Host of the popular The Skies We’re Under Podcast, Rachel’s a passionate speaker, trainer, qualified nurse and unqualified mum of three; one who has severe disabilities, life-limiting epilepsy and an addiction to the gameshow Pointless. An expert through lived experience, Rachel leads Born at the Right Time to support families of people with complex disabilities whilst also Bridging the Gap between them and the practitioners who support them through CPD certified training, influencing policy makers and turning rhetoric into reality.

Essentials in Communication and Co-production with Parents/Carers (Sept 24)
5.0(1)By Born at the Right Time
Inspiring, interactive and unique 4-hour CPD certified training on Communication and Co-production with Parents/Carers

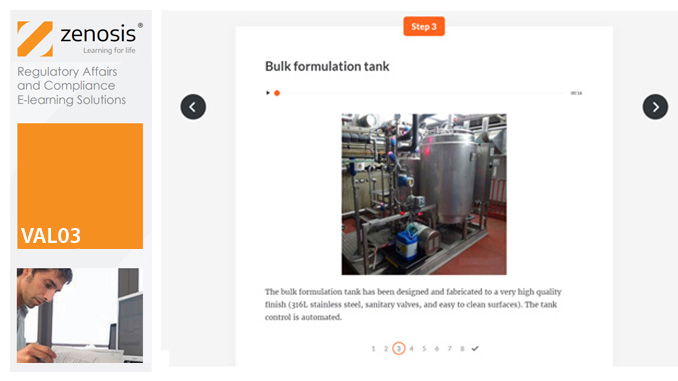
VAL03: Commissioning and Installation Qualification
By Zenosis
Before equipment can be used routinely in production, it must first be commissioned and, if necessary, undergo Installation Qualification (IQ). This module describes commissioning and IQ requirements and procedures in the medicines and healthcare products industries. It follows the activities of a typical validation team as they carry out a project for a pharmaceutical company.
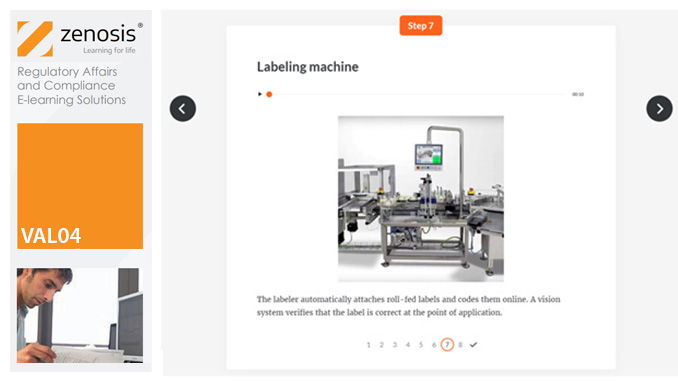
VAL04: Operational and Performance Qualification
By Zenosis
Having undergone Installation Qualification, before equipment can be used routinely in production, it needs to undergo Operational Qualification (OQ) and Performance Qualification (PQ). This module describes OQ and PQ requirements and procedures in the medicines and healthcare products industries. It follows the activities of a typical validation team as they carry out a project for a pharmaceutical company.
A Beginner’s Guide to Photographing Jewellery Saturday 10th August 10.00 – 3.30pm
5.0(21)By Workshop 925

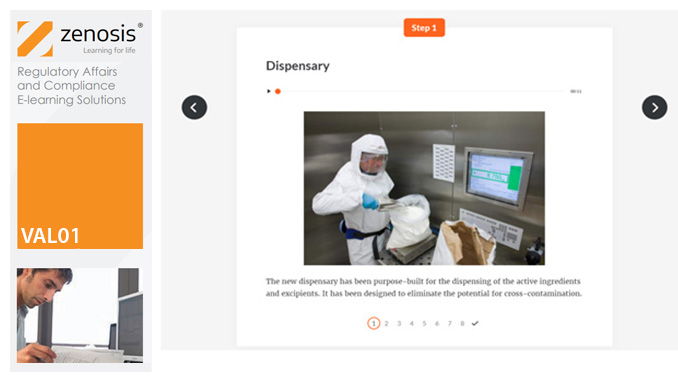
VAL01: Introduction to Validation
By Zenosis
Validation of equipment, services, systems and processes is vitally important in the medicines and healthcare products industries. Regulatory authorities require documented evidence that manufacturing processes will consistently result in products meeting predetermined quality standards. This module provides an introduction to validation and to the regulations and guidance that apply to it. It describes the activities of a typical validation team as they carry out a project for a pharmaceutical company.
Search By Location
- EQ Courses in London
- EQ Courses in Birmingham
- EQ Courses in Glasgow
- EQ Courses in Liverpool
- EQ Courses in Bristol
- EQ Courses in Manchester
- EQ Courses in Sheffield
- EQ Courses in Leeds
- EQ Courses in Edinburgh
- EQ Courses in Leicester
- EQ Courses in Coventry
- EQ Courses in Bradford
- EQ Courses in Cardiff
- EQ Courses in Belfast
- EQ Courses in Nottingham



