- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
2324 Courses
Cognitive Stimulation Therapy (CST) Training online via Zoom
By Dementia Pathfinders Community Interest Company
This one day training will give learners an overview of how to deliver Cognitive Stimulation Therapy (CST). CST is a brief evidence-based group treatment for people with dementia. This interactive course will include a background about CST and how it can be used, research overview and detailed presentation of the treatment, including video footage and role-play.

Clinical Coding & Billing (Medical Coding) Course
By Training Express

Level 5 Diploma in Medical & Clinical Administration
By Training Tale

Clinical Governance in Adult Care
By Apex Learning

Medical Writing, Medical Coding & Medical Terminology(Clinical Coding) - CPD Certified
4.8(9)By Skill Up
CPD Certified | 7-in-1 Premium Bundle | **Gift: 1 Free PDF Certificate | High-quality Materials | 24/7 Learner Support

CT04a - Clinical trials in drug development
By Zenosis
New drug development requires major investment in capital, human resources and technical expertise. Strict adherence to regulations on testing and manufacturing standards is also required before a new drug can be marketed. One of the greatest challenges in conducting clinical trials is that of efficiency. As trials become more comprehensive, involving large numbers of participants globally, their duration is prolonged and costs increase. The longer trials last, the shorter is the patent life remaining after market approval and the longer patients must wait for the new product. This short course covers the key components of clinical trials and how these requirements interact with the drug development cycle.

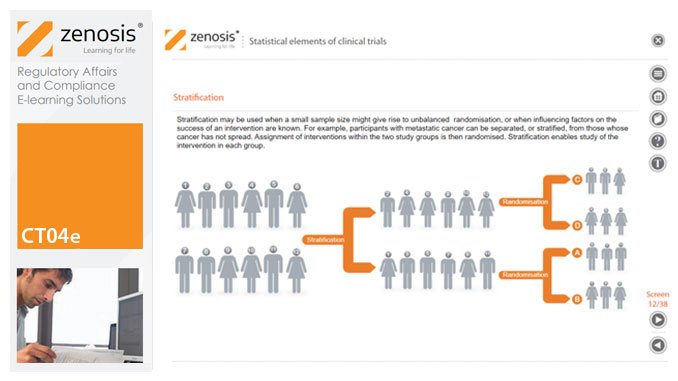
CT04e - Statistical elements of clinical trials
By Zenosis
Analytical statistical elements are essential concepts in the design of clinical trials. This analysis helps us to understand whether a conclusion from a study of a sample of the target population applies generally to that population as a whole. In particular, it helps us to answer the question: Did the treatment effect in the given study occur just by chance? The statistical elements of a well-controlled study minimise the chances of drawing the wrong conclusions, by providing clear thresholds for such errors. The basic statistical elements of a clinical trial include eligibility criteria, randomisation, sample size, power, and blinding, and these are discussed in this short course.
Search By Location
- Clinical Courses in London
- Clinical Courses in Birmingham
- Clinical Courses in Glasgow
- Clinical Courses in Liverpool
- Clinical Courses in Bristol
- Clinical Courses in Manchester
- Clinical Courses in Sheffield
- Clinical Courses in Leeds
- Clinical Courses in Edinburgh
- Clinical Courses in Leicester
- Clinical Courses in Coventry
- Clinical Courses in Bradford
- Clinical Courses in Cardiff
- Clinical Courses in Belfast
- Clinical Courses in Nottingham


