- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
2324 Courses
CT03e - Clinical trial investigator’s GCP responsibilities
By Zenosis
A clinical investigator is responsible for conducting the clinical trial in compliance with the study protocol, GCP, medical ethics, and applicable legal requirements. The clinical research community expects that investigators and clinical staff are fully trained in GCP. Duties and functions discussed in this short course include: provision of adequate resources; liaison with IRB/IEC; compliance with protocol; management of investigational product(s), informed consent and data records; and safety reporting.
Unlock the secrets of effective and sustainable weight loss with our comprehensive course, 'Clinical Weight Loss: Strategies and Techniques.' Explore the intersection of nutrition, exercise, behavioral psychology, and advanced clinical approaches to address obesity. Equip yourself with evidence-based practices, personalized interventions, and the multidisciplinary skills needed to guide individuals on successful weight loss journeys. Join us for a transformative learning experience in healthcare, blending science and practical application for improved patient outcomes.

Medical & Clinical Administration Course
By Training Express

Clinical cupping therapy course Course
By NextGen Learning

Nursing Assistant, Phlebotomy & Clinical Observation
By NextGen Learning

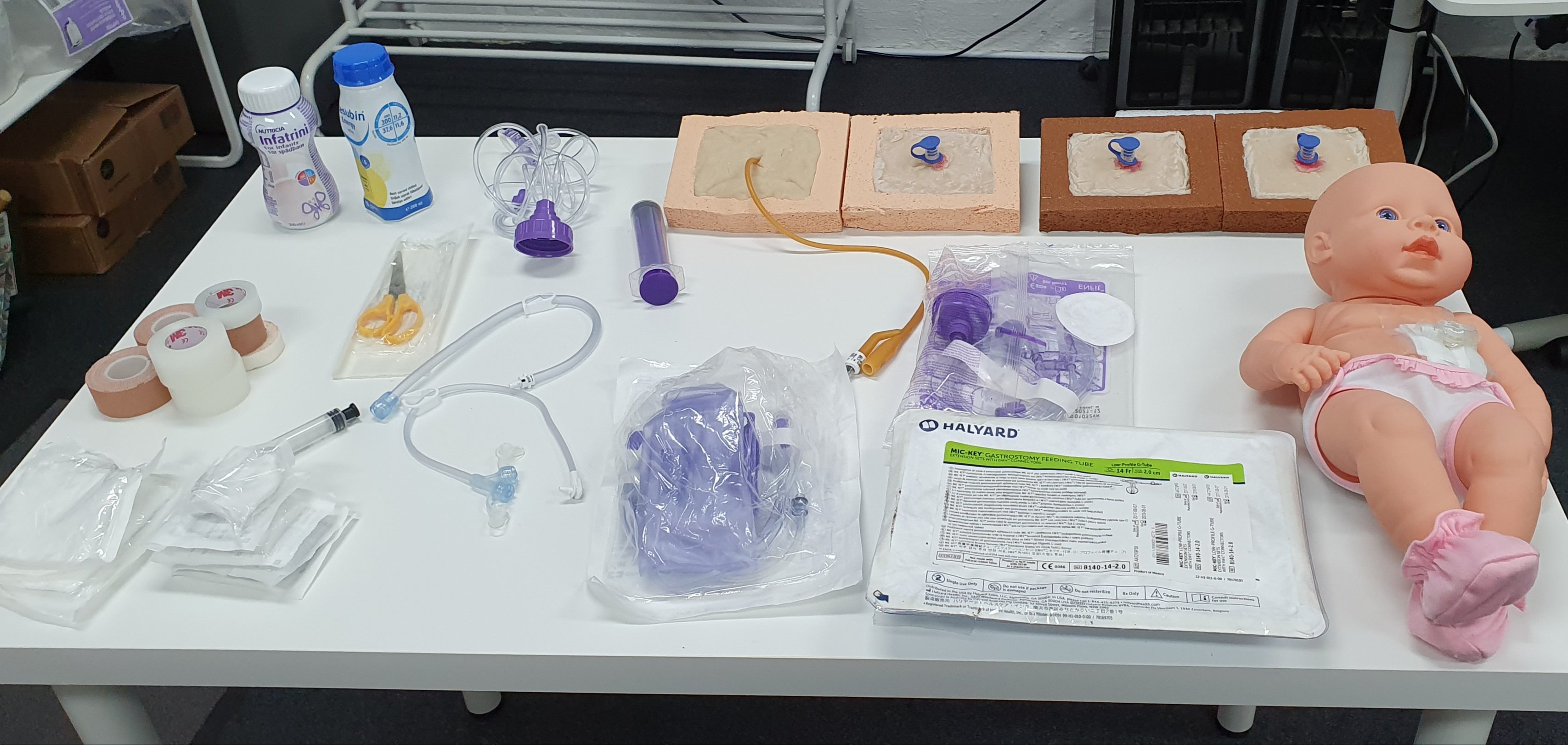
Gastrostomy tube care Gastrostomy tube management Gastrostomy tube training Enteral feeding care Gastrostomy tube complications Gastrostomy site care Gastrostomy tube insertion Gastrostomy tube feeding techniques Gastrostomy tube nursing Gastrostomy care for healthcare professionals Gastrostomy tube education CPD accredited course Nursing revalidation hours Practical gastrostomy care training Hands-on gastrostomy tube practice Patient education in gastrostomy care Ethical considerations in gastrostomy tube care Cultural sensitivity in gastrostomy care Gastrostomy tube complications prevention High-quality gastrostomy care certification PEG
Medical and Clinical Administration Course
By Lead Academy

Search By Location
- Clinical Courses in London
- Clinical Courses in Birmingham
- Clinical Courses in Glasgow
- Clinical Courses in Liverpool
- Clinical Courses in Bristol
- Clinical Courses in Manchester
- Clinical Courses in Sheffield
- Clinical Courses in Leeds
- Clinical Courses in Edinburgh
- Clinical Courses in Leicester
- Clinical Courses in Coventry
- Clinical Courses in Bradford
- Clinical Courses in Cardiff
- Clinical Courses in Belfast
- Clinical Courses in Nottingham

