- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
2324 Courses
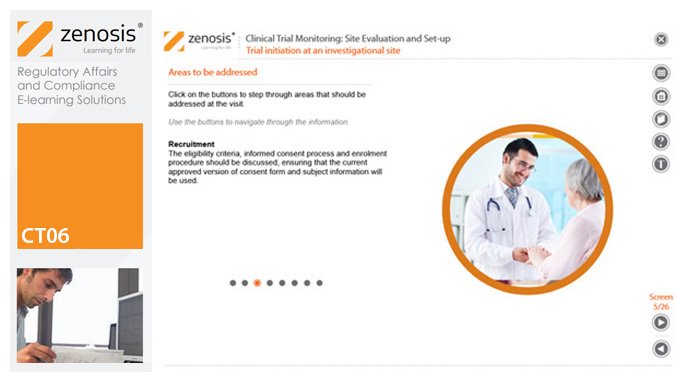
CT06: Clinical Trial Monitoring: Site Evaluation and Setup
By Zenosis
The sponsor of a clinical trial needs to reach agreement with clinical investigators to conduct the trial. The suitability of investigators and their institutional sites, typically hospitals, has to be evaluated, and the trial has to be set up at each site. This module describes the processes involved, focusing particularly on the role of a Clinical Research Associate (CRA) employed or contracted by the sponsor to monitor the trial.
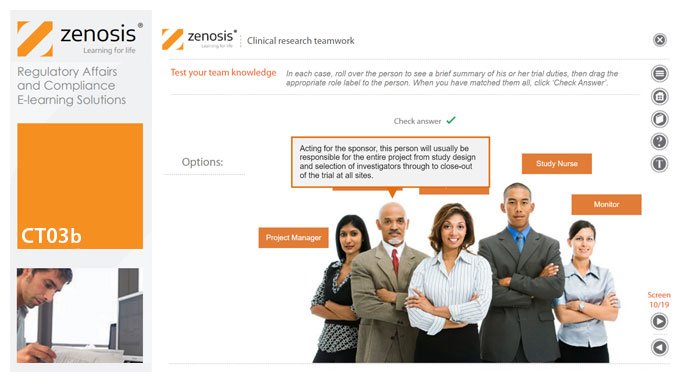
CT03b - Clinical research teamwork
By Zenosis
A clinical trial, particularly a late-phase commercial study, is a major project requiring collaboration between the sponsor and staff or contractor, on the one hand, and the clinical investigator(s) and other healthcare professionals on the other. Good communication among all parties is essential. In this short course we introduce the major roles in a typical clinical research project and outline their duties.
Clinical Anaesthesia Training
By Online Training Academy

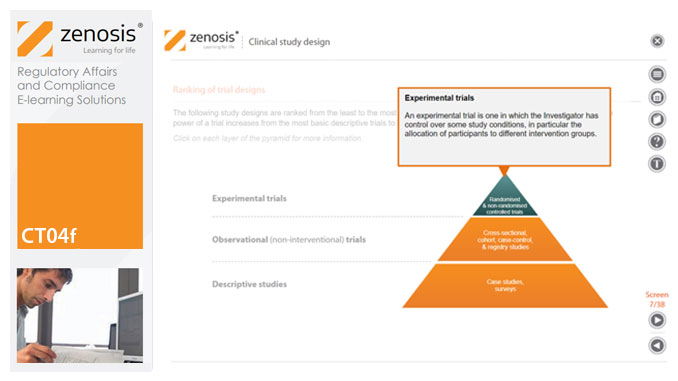
CT04f - Clinical study design
By Zenosis
Clinical trial design establishes the framework upon which the clinical trial process will be conducted, and sets the objectives of the trial. The application for marketing approval, submitted to the regulatory authorities, will provide clinical data reflecting the trial design. Since trial design impacts the whole drug development process and lifecycle, particular care and due diligence is essential. This short course provides an overview of the main types of study design.
CT09: Good Clinical Practice Inspections and Audits
By Zenosis
The module describes general principles of GCP inspection and audit, discusses preparation for an inspection, and sets out in detail what European and US FDA inspectors will examine. Finally it describes post-inspection actions by the regulator and the inspected party.

Trauma Informed Practice in Education is a research evidence based whole school systems approach that starts with you. Improved attendance, improved learning outcomes, reduced suspensions and exclusions and better staff recruitment, retention and wellbeing all begin from a place of understanding child development, the impact of adversity on child development and the role of us as educators in supporting opportunities for growth and resilience. Systems change takes teamwork, however the most important member of that team is you. On this Trauma Informed Practice Day you will learn the why and how to keep you well and resourced in order to support the emotional and academic progress of the children in your school. You may already have some knowledge or you may be coming as a complete novice. Either and anything in between is fine. Numbers are limited to keep the group small for the benefit of more enriched learning. On this practice day you will; • Take a deep learning dive into the nervous system and how this applies to you, your setting and the progress of your learners. • Learn practical skills and strategies to support yourself and others. • Have opportunities to discuss children you are working with through small group supervision sessions • Benefit from a day of immersion into the topic with two highly qualified and experienced practitioners.

CT04: An Introduction to Clinical Trial Preparation and Design
By Zenosis
This module aims to provide you with effective strategies for the preparation and conduct of a clinical trial, while adhering to regulatory safety standards. Management of data for submission is also covered.
Search By Location
- Clinical Courses in London
- Clinical Courses in Birmingham
- Clinical Courses in Glasgow
- Clinical Courses in Liverpool
- Clinical Courses in Bristol
- Clinical Courses in Manchester
- Clinical Courses in Sheffield
- Clinical Courses in Leeds
- Clinical Courses in Edinburgh
- Clinical Courses in Leicester
- Clinical Courses in Coventry
- Clinical Courses in Bradford
- Clinical Courses in Cardiff
- Clinical Courses in Belfast
- Clinical Courses in Nottingham


