- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
2920 Courses
Level 3 & 5 Proofreading Course at QLS
By Imperial Academy
Level 5 QLS Endorsed Course | Endorsed Certificate Included | Plus 5 Career Guided Courses | CPD Accredited

Project Manager's Fast Start Program
By OnlinePMCourses
Start your Journey to Becoming a Successful Project Manager, without waiting for other People to See your Potential

Oracle Database Administration from Zero to Hero
By Packt
In this comprehensive course, we will guide you through the world of Oracle Database Administration, starting from the basics and equipping you with the skills to create databases, manage storage, implement backup plans, apply patches, troubleshoot issues, and excel in the role of a professional DBA.

SUB03: The Mutual Recognition Procedure (MRP)
By Zenosis
his module describes the roles of the various players in the procedure, the sequence and duration of the stages involved, and the requirements on content, format and timing of submissions. It discusses the special issues that apply to generic products in the MRP.
SUB11: The Decentralised Procedure (DCP)
By Zenosis
This module describes the roles of the various players in the procedure, the sequence and duration of the stages involved, and the requirements on content, format and timing of submissions. It discusses the special issues that apply to generic products in the DCP.
Web Development Secrets Revealed - Critical Rendering Path, HTTP, AJAX, and More
By Packt
A comprehensive web development course that will help you understand 'why' things work and not just 'how'. Learn to write better code to boost your website traffic; know how to precision fix and tweak behavior and performance; improve your market penetration and your margins. Everything you need to know about the Critical Rendering Path, AJAX, and HTTP is right here at your fingertips.

SUB14: The Regulatory Pathway to Licensure of Follow-on Biologics (Biosimilars) in the USA
By Zenosis
The regulation of biological medicinal products is governed by different laws from those that apply to small-molecule synthetic drugs. Producing faithful copies of therapeutic proteins is more challenging than producing generic drugs. The US legal framework for the licensure of follow-on biologics, and accompanying regulatory guidance from the Food and Drug Administration (FDA), have been established only in recent years.

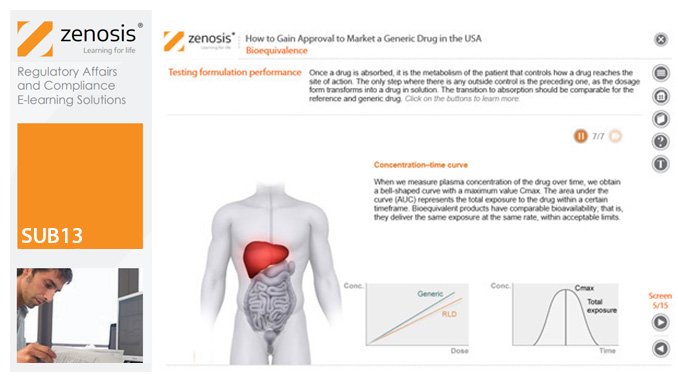
SUB13: How to Gain Approval to Market a Generic Drug in the USA
By Zenosis
This module outlines the legislative and regulatory context for the development of generic drugs and describes the essential role of the Abbreviated New Drug Application (ANDA) in gaining marketing approval. The use of information in the ‘Orange Book’ is explained, as is the role of patent certification in the application. The importance of establishing bioequivalence between a generic and its reference product is emphasised. The module specifies the content and format requirements for an ANDA submission and describes the FDA’s review and approval process. An outline is given of the Generic Drug User Fee Amendments (GDUFA) and the law’s effects on industry players.
Search By Location
- author Courses in London
- author Courses in Birmingham
- author Courses in Glasgow
- author Courses in Liverpool
- author Courses in Bristol
- author Courses in Manchester
- author Courses in Sheffield
- author Courses in Leeds
- author Courses in Edinburgh
- author Courses in Leicester
- author Courses in Coventry
- author Courses in Bradford
- author Courses in Cardiff
- author Courses in Belfast
- author Courses in Nottingham

