- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
7385 Courses
Safety Harness Awareness
By Vp ESS Training
This ESS course is suitable for operatives who during the daily activities have to wear a safety harness. It will give the delegate an understanding of the safe use of a harness, methods of inspection and awareness of the regulations. Book via our website @ ESS | Working at Heights training | Vp ESS (vp-ess.com) or via email at: esstrainingsales@vpplc.com or phone on: 0800 000 346

Safe Use of Ladders and Stepladders
By Vp ESS Training
This ESS course is suitable for operatives who during the daily activities have to use Ladders. It will give the delegate an understanding of the safe use and inspection as well as awareness of the regulations. Book via our website @ ESS | Working at Heights training | Vp ESS (vp-ess.com) or via email at: esstrainingsales@vpplc.com or phone on: 0800 000 346

Safe Use of Hand Held Power Tools
By Vp ESS Training
This ESS course is suitable for operatives who during the daily activities have to use power tools. By the end of the course delegates will have an understanding of the correct operational procedures of power tools. Book via our website @ ESS | Training Courses | Vp ESS (vp-ess.com) or via email at: esstrainingsales@vpplc.com or phone on: 0800 000 346

SMSTS (Site Management Safety Training Scheme)
By 2020 Training
This five-day Site management safety training scheme (SMSTS) course is designed for site managers, agents and persons who are, or are about to be, responsible for planning, organising, monitoring, controlling and administering groups of staff within a construction environment. The course covers all relevant legislation and other aspects which affect safe working in the building, construction and civil engineering industries. It highlights the need for risk assessment in the workplace, the implementation of the necessary control measures, and adequate communication to sustain a health and safety culture among the workforce. Delivered interactively, the course includes group and individual exercises, discussions and projects.

Fraud Prevention Strategies and Essential Fraud Awareness
By NextGen Learning

GDPR in Healthcare Training
By Course Gate
Gain a comprehensive understanding of GDPR compliance in healthcare with our GDPR for Health and Social Care course. Learn to handle personal data securely and uphold patient confidentiality while meeting legal requirements. Ideal for healthcare and social care providers, this course covers GDPR essentials, data security awareness, and more.

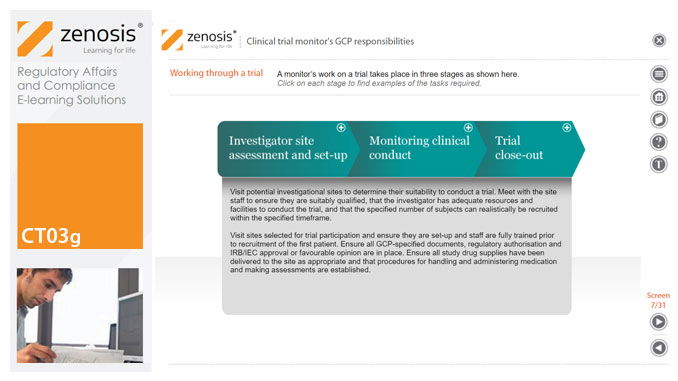
CT03g - Clinical trial monitor’s GCP responsibilities
By Zenosis
A clinical trial monitor acts on behalf of the sponsor to support investigational site personnel, verify the accuracy of data recorded, and ensure that the trial is conducted in compliance with the protocol, GCP and other study specific requirements. He or she acts as the ‘eyes and ears’ of the sponsor at the investigational site and provides the main channel of communication between sponsor and investigator. This short course explores the responsibilities of the monitor and provides insight into key challenges. We discuss assessment of investigators and investigational sites, education and trial initiation, monitoring of clinical conduct, including CRF review and source document verification, and trial close-out. We discuss noncompliance and how to deal with it.
Understanding Workplace Bullying and Harassment (In-house)
By Conduct Change Ltd
Understanding Workplace Bullying and Harassment The definitive guide to workplace bullying & harassment delivered by UK's leading expert.

Search By Location
- Compliance Courses in London
- Compliance Courses in Birmingham
- Compliance Courses in Glasgow
- Compliance Courses in Liverpool
- Compliance Courses in Bristol
- Compliance Courses in Manchester
- Compliance Courses in Sheffield
- Compliance Courses in Leeds
- Compliance Courses in Edinburgh
- Compliance Courses in Leicester
- Compliance Courses in Coventry
- Compliance Courses in Bradford
- Compliance Courses in Cardiff
- Compliance Courses in Belfast
- Compliance Courses in Nottingham

