- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
3952 Courses delivered Online
Red Hat RHCSA Rapid Track Course (RH199)
By Nexus Human

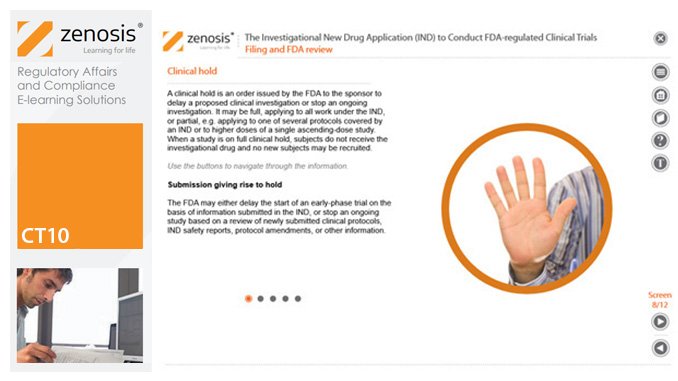
CT10: The Investigational New Drug Application (IND) to Conduct FDA-regulated Clinical Trials
By Zenosis
An Investigational New Drug Application (IND) is a submission to the US Food and Drug Administration (FDA) for permission to conduct a clinical trial of a medicinal product. This module describes regulatory requirements that sponsors or sponsor-investigators must meet for successful compilation, filing and maintenance of INDs. The IND and its role are defined, and the contexts in which it is required are specified.
Secretary PA Training
By EduTech - Taylor Mason
During this 2 day course, you will develop a learning-based action plan to use in your workplace ensuring that you can put the learning into action.

Secretary PA Training
By EduTech - Taylor Mason
During this 2 day course, you will develop a learning-based action plan to use in your workplace ensuring that you can put the learning into action.

Secretary PA Training
By EduTech - Taylor Mason
During this 2 day course, you will develop a learning-based action plan to use in your workplace ensuring that you can put the learning into action.

Nursing, Clinical Observations with Occupational Therapy
By Apex Learning

Ayurveda, Homeopathy & Safe Handling of Medicines
By Imperial Academy
Level 5 - Three QLS Endorsed Diploma | QLS Hard Copy Certificate Included | Plus 10 CPD Courses | Lifetime Access



