- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
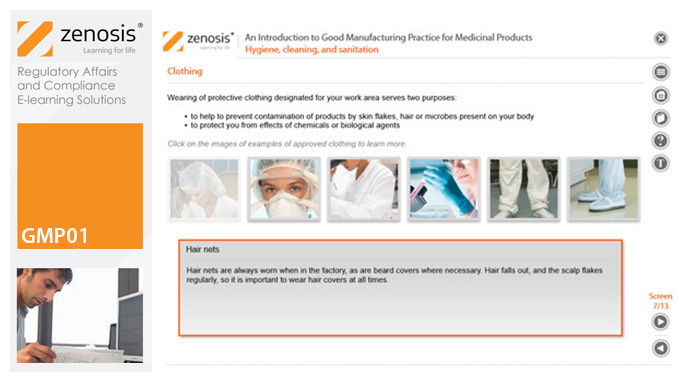
GMP01: An Introduction to Good Manufacturing Practice for Medicinal Products
By Zenosis
Good Manufacturing Practice (GMP) is a set of rules for medicines manufacturers to follow so that their products are safe, effective, and of good quality. The rules may be written into law or set out in guidance documents from regulatory authorities. Regulators will not allow medicinal products to be placed, or to remain, on the market in their country unless the products can be shown to be manufactured in compliance with GMP. To this end, they carry out inspections of manufacturing plants. Companies that persistently commit serious breaches of GMP requirements have suffered huge fines.
Merchandising - 30 CPD Courses Bundle
By Compliance Central

Logistic Management - 30 CPD Courses Bundle
By Compliance Central

IATP - Asbestos Awareness Online
By Airborne Environmental Consultants Ltd
If you have no intention of removing asbestos but work on buildings built or refurbished before the year 2000, asbestos could be present. You will need awareness training so you know how to avoid the risks. Asbestos awareness training should be given to employees whose work could foreseeably disturb the fabric of a building and expose them to asbestos or who supervise or influence the work. In particular, it should be given to those workers in the refurbishment, maintenance and allied trades where it is foreseeable that ACMs may become exposed during their work.






