- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
549 Courses delivered Online
Certified TIA-942 Design Consultant (CTDC)
By Nexus Human

Project Management Fundamentals for IT Projects: Virtual In-House Training
By IIL Europe Ltd

Licensed Premises Staff Training-CPD & Institute of Hospitality Approved
By BAB Business Group
The alcohol licensing laws of the United Kingdom regulate the sale and consumption of alcohol. It is important for anyone working in licensed premises to have a basic understanding of these laws and how they apply to them. This course starts with an overview of UK alcohol licensing laws and the importance of the four key licensing objectives. The course will also discuss mandatory conditions, which are compulsory for all licensed premises and looks in detail at the major issue of age verification. Finally, it outlines the responsibilities

Carbon Capture, Utilization & Storage (CCUS)
By EnergyEdge - Training for a Sustainable Energy Future

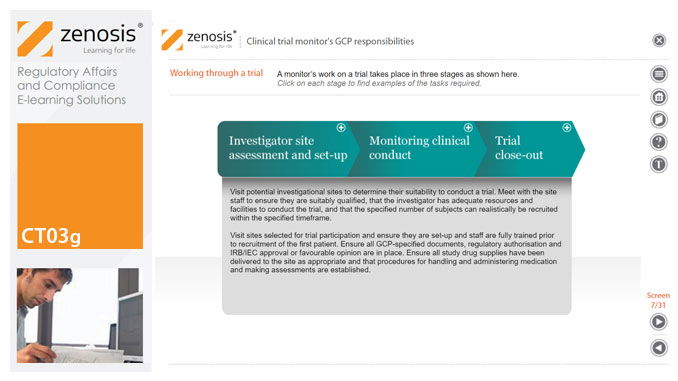
CT03g - Clinical trial monitor’s GCP responsibilities
By Zenosis
A clinical trial monitor acts on behalf of the sponsor to support investigational site personnel, verify the accuracy of data recorded, and ensure that the trial is conducted in compliance with the protocol, GCP and other study specific requirements. He or she acts as the ‘eyes and ears’ of the sponsor at the investigational site and provides the main channel of communication between sponsor and investigator. This short course explores the responsibilities of the monitor and provides insight into key challenges. We discuss assessment of investigators and investigational sites, education and trial initiation, monitoring of clinical conduct, including CRF review and source document verification, and trial close-out. We discuss noncompliance and how to deal with it.
Carbon Capture and Storage (CCS): Project Risks & How to Manage Them
By EnergyEdge - Training for a Sustainable Energy Future


