- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
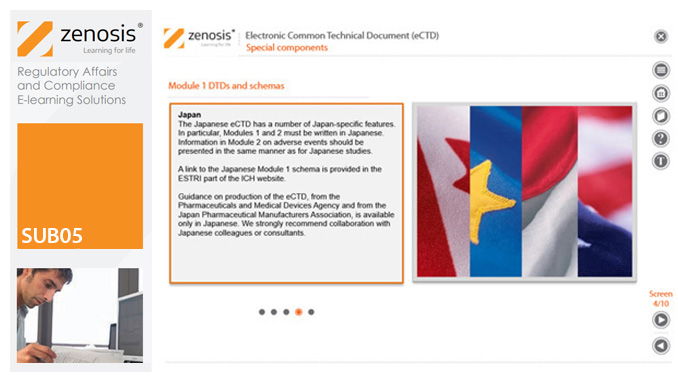
SUB05: Electronic Common Technical Document (eCTD)
By Zenosis
The eCTD is mandatory for all applications for marketing approval and all subsequent related submissions in the European Economic Area, the USA and Canada. Other countries intend to make its use mandatory. The eCTD specification has been developed to facilitate the global electronic submission, review and lifecycle management of medicinal product dossiers for regulatory applications. It broadens the scope of the CTD to include information on variations, renewals and amendments, so that it is no longer a static document but is updatable throughout the life of the product. This module outlines the eCTD specification, discusses the approach to regional differences in dossiers, and provides guidance on creation of an eCTD submission. The module provides a training and reference tool that will be of particular value to those new to the use of the format.
EYFS and Lesson Planning for Teaching - QLS Endorsed Certificate
By Imperial Academy
Level 3 & 5 Endorsed Diploma | QLS Hard Copy Certificate Included | Plus 5 CPD Courses | Lifetime Access

Level 3 Teaching Assistant Award with School Administration & Management Training
By Kingston Open College
Premium Bundle of all Time | Ofqual Regulation + NCFE Awards + CPD Accreditation | Assessment & Tutor Support Included

Level 1 Essential Digital Skills Qualification - (EDSQ)
By Lead Academy

Sports Therapy and Coaching with Sports Psychology - 3 in 1 QLS Bundle
By Imperial Academy
3 QLS Endorsed Diploma | QLS Hard Copy Certificate Included | 10 CPD Courses with Certificates | Installment Options