- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
4478 Courses delivered Online
CT03a - ICH, harmonisation, and principles of Good Clinical Practice
By Zenosis
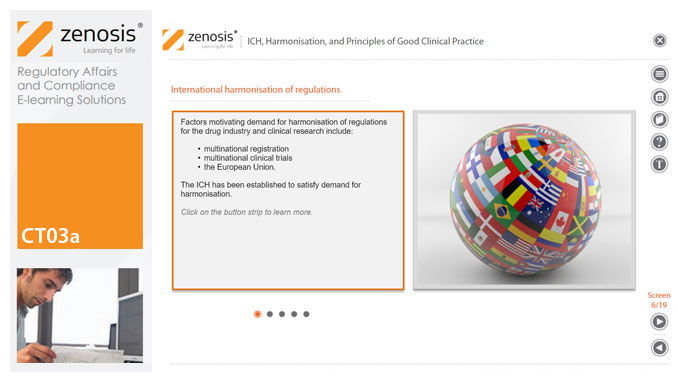
Good Clinical Practice (GCP) is a set of internationally recognised ethical and scientific quality requirements for designing, conducting, recording and reporting clinical trials. Compliance with GCP principles is required by regulatory authorities in many countries for the authorisation of clinical trials and the acceptance of their data in applications for marketing approval. The International Council for Harmonisation's guideline E6, often referred to as ICH GCP, is the international standard specification for Good Clinical Practice. In this short course we describe the ICH’s role in the harmonisation of regulations, introduce its guideline E6, and set out the principles of GCP.
Diploma in Sustainable Energy- Level 5 QLS Endorsed Course
By One Education

Business Finance - QLS Endorsed Certificate
By Imperial Academy
5 QLS Endorsed Course With Certificates | 5-in-1 Bundle | CPD Accredited | Career Guided Program | Lifetime Access

Level 5 QLS Diploma in Construction Site Management & Cost Estimation
By Imperial Academy
Optimize construction projects with efficient site management and accurate cost estimation for success in the UK market

Air Pollution Management And Control Course
By Elearncollege

Anti Money Laundering (AML) - QLS Level 5 Endorsed Course with Certificate
By Imperial Academy
Level 5 QLS Endorsed Certificate Included | 150 CPD Points | Lifetime Access | Installment Payment | Tutor Support

Payroll, Sage 50, Tax for Payroll Managers
By Apex Learning

Food Labelling and Food Safety - Double Endorsed Certificate
By Imperial Academy
2 QLS Endorsed Course | CPD Certified | Free PDF + Hardcopy Certificates | 80 CPD Points | Lifetime Access


