- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
Care Assistant Career Track Diploma Course
By Training Express

Injection Technique and Immunisation Training for HCAs
By M&K Update Ltd
A two-day Immunisation Training Course for Healthcare Support Workers/Healthcare Assistants (HCSWs/HCAs).

Medical Coding Inpatient Training: DRG-PCS
By Apex Learning

DNA , Genealogy & Laboratory Technician - Double Endorsed Certificate
By Imperial Academy
2 QLS Endorsed Course | CPD Certified | Free PDF + Hardcopy Certificates | 80 CPD Points | Lifetime Access

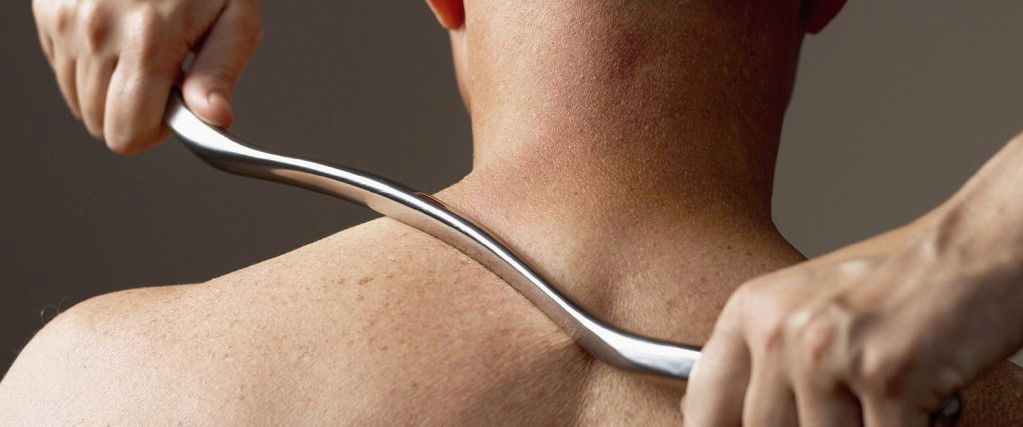
IASTM (Instrument Assisted Soft Tissue Mobilisation)
5.0(8)By The Medical Massage Lady Training School
This course will introduce you to IASTM and help you to select specific tools, techniques and treatment plans for clinical conditions, and identify indications and contraindications of IASTM. You will be able to explain the reasoning behind IASTM and compare mechanical and physiological responses of IASTM. You will also learn the difference between a fascial restriction and muscle tightness and how to identify common fascial restrictions.There is also information to help you differentiate between similar conditions and to differentiate between active and latent trigger points.
PV03: Drug Safety and Pharmacovigilance
By Zenosis
Drug safety monitoring and risk management are vitally important for medicinal product developers, licence holders and clinical investigators. In addition to their duty to protect public health, increasingly tight regulation and potentially massive payments to litigants provide strong incentives for pharmaceutical and biotechnology companies to ensure that they maintain efficient systems for drug safety / pharmacovigilance and that all staff are aware of the basic requirements. This course will provide them with an overview of the most important aspects of this discipline, both before and after marketing of products, especially as they apply in Europe and the USA.

Evidence Based Health Care Course
By Elearncollege

Principles of Infection Prevention and Control Level 2 - QLS Endorsed Course
By One Education

Principles of Infection Prevention and Control Level 2 - QLS Endorsed Course
By One Education

Level 5 Diploma in Health & Social Care - QLS Endorsed
By Compliance Central
