- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
Drug and Alcohol Awareness Training - CPD Accredited
By Training Tale

QUALIFI Level 3 Diploma in Data Science
By School of Business and Technology London

GLP02 - Good Quality Control Laboratory Practice
By Zenosis
The medicinal products industry is heavily regulated by governments. Within the industry’s Good Manufacturing Practice (GMP) framework, analytical laboratories engaged in quality control (QC) of starting materials, intermediates, bulk products, finished products, and packaging need to comply with relevant GMP standards. We refer to these as Good Quality Control Laboratory Practice, or GQCLP. Regulatory authorities inspect laboratories to confirm that they meet the standards. This course explains how to comply with GQCLP, and it provides advice on laboratory work in general.

Working in Confined Spaces-IIRSM, CPD & IOSH Approved
By BAB Business Group
At the end of this course you’ll be able to demonstrate your understanding of the control measures to be followed when working in, or near to, confined spaces as detailed in a safe working procedure This course will cover the legislation associates with working in confined spaces. What counts as a confined space, the potential hazards, safe operating procedures, emergency procedures and rescue. Important note: This is an awareness course only, designed for people who need to be aware of the hazards and risks of confined space working but are not required to enter a confined space. If you are required to perform any work activity in, or in the proximity of, a confined space then you will also need to have an 'approved' standard of practical training at the 'appropriate' level.

HR Management (Crisis Communication, Conflict & Stress Management)
4.8(12)By Academy for Health and Fitness

Ofqual Regulated Level 3 Teaching Assistant Award with Preschool Teaching
By Kingston Open College
Premium Bundle of all Time | Ofqual Regulation + NCFE Awards + CPD Accreditation | Assessment & Tutor Support Included

Paediatric First Aid for Teaching Assistants
By The Teachers Training

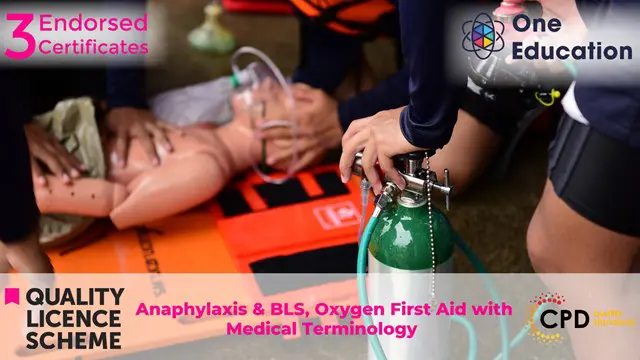
Anaphylaxis & BLS, Oxygen First Aid wth Medical Terminology Course
By One Education
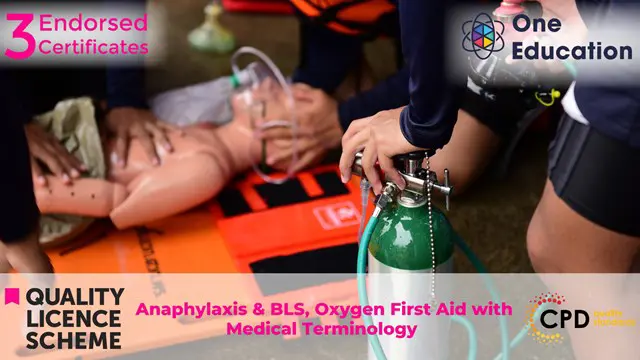
Anaphylaxis & BLS, Oxygen First Aid wth Medical Terminology Course
By One Education
Dog Cleaning, Dog Care and Grooming with Dog First Aid Course
By One Education
