- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
1204 Courses delivered Online
Master the art of effective data management with our 'Data Documentation and Record Keeping Essentials' course. Explore key topics including data collection, storage, and security, while developing strategies for comprehensive documentation. Gain the skills needed for compliance, access control, and successful record-keeping. Elevate your professional profile with essential data skills and contribute to organizational success. Enroll now for a transformative learning experience in the world of data management.

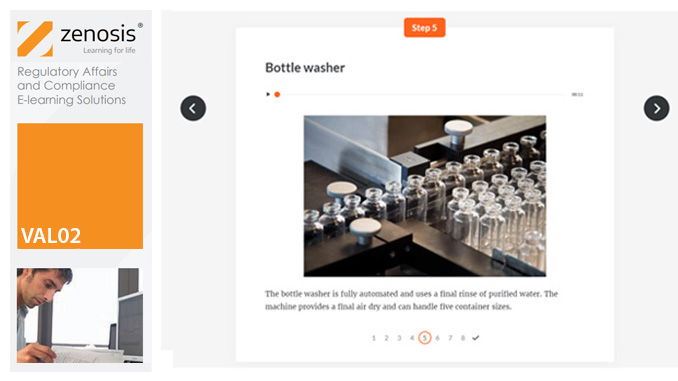
VAL02: Validation Plans and Documentation
By Zenosis
Essential to validation is the provision of documented evidence verifying that manufacturing processes will consistently result in products meeting predetermined quality standards. This module describes the purpose, content and use of validation master plans, project validation plans, and other documentation for validation projects in the medicines and healthcare products industries. It describes the activities of a typical validation team as they carry out a project for a pharmaceutical company.
C20M01 - Intruder & Hold up alarm foundation EN50131 (online)
By Ember Compliance
Learners will be introduced to the design, installation, commissioning, and maintenance of intruder and hold-up alarm systems.

Creating API Documentation and Developer Portals Course
By Cherryleaf

CT03c - Clinical trial documentation
By Zenosis
Regulatory authorities tend to abide by the maxim that ‘If it isn’t documented, it didn’t happen’. Rigorous documentation of all aspects of a clinical trial is necessary to provide evidence of GCP and compliance with regulatory requirements, as well as enabling effective management of the trial. In this short course we describe important examples of the documents designated by ICH GCP as essential to the conduct of a clinical trial.

Introduction to Computer Systems Validation
By Research Quality Association

CompTIA Cybersecurity Analyst (CySA+)
By Nexus Human

Platelet-rich Plasma (PRP) treatments Nationally Recognised Qualification No previous experience or qualifications needed Open College Network Accreditation Level 4 (as required for minimally invasive procedures) Covers standards set by HEE Employed (salon) or Self-Employed opportunities Basic understanding of English language required OPEN TO ALL APPLICANTS