- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
Xero Accounting and Bookkeeping at QLS Level 7 Diploma - 12 Courses Bundle
By NextGen Learning

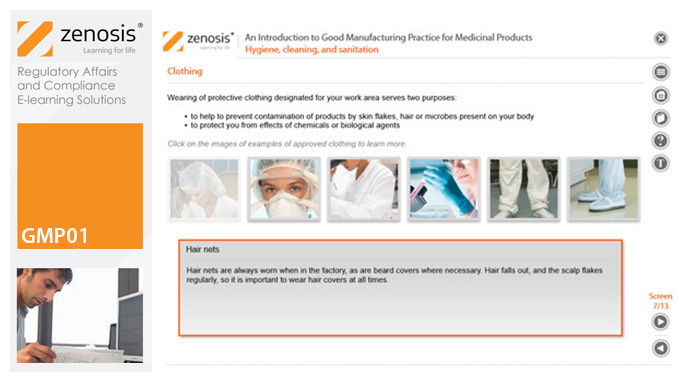
GMP01: An Introduction to Good Manufacturing Practice for Medicinal Products
By Zenosis
Good Manufacturing Practice (GMP) is a set of rules for medicines manufacturers to follow so that their products are safe, effective, and of good quality. The rules may be written into law or set out in guidance documents from regulatory authorities. Regulators will not allow medicinal products to be placed, or to remain, on the market in their country unless the products can be shown to be manufactured in compliance with GMP. To this end, they carry out inspections of manufacturing plants. Companies that persistently commit serious breaches of GMP requirements have suffered huge fines.
General Data Protection Regulation Foundation (GDPR F)
By Packt
Implement and manage a compliance framework to protect personal data using GDPR Foundation training.

Master the art of safeguarding your business with our Transaction Monitoring Masterclass. Detect and prevent fraud effectively through in-depth exploration of regulatory frameworks, risk-based approaches, advanced monitoring systems, and the future of transaction security. Join us to acquire essential skills, reduce false positives, and stay ahead in the ever-evolving landscape of financial security. Protect your business with confidence.
Fashion Design Essentials Course
By One Education

Fashion Design Essentials Course
By One Education

Property Sales, Negotiation with Tenant Management
By Apex Learning



