- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
11702 Courses delivered Online
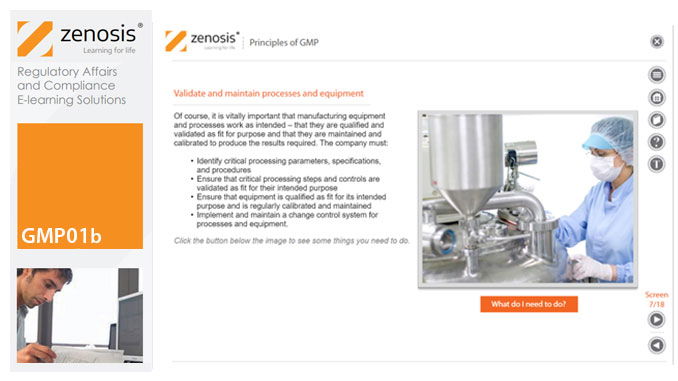
GMP01b - Principles of GMP
By Zenosis
In this short course we present an overview of the main principles of GMP, and we outline some things that manufacturing personnel need to do to comply with requirements. We identify the principal goals of GMP as: prevention of contamination; prevention of mix-ups; scrupulous documentation; validation and maintenance of processes and equipment; quality assurance by an independent unit; and training. We place GMP in the context of a company’s quality management system.
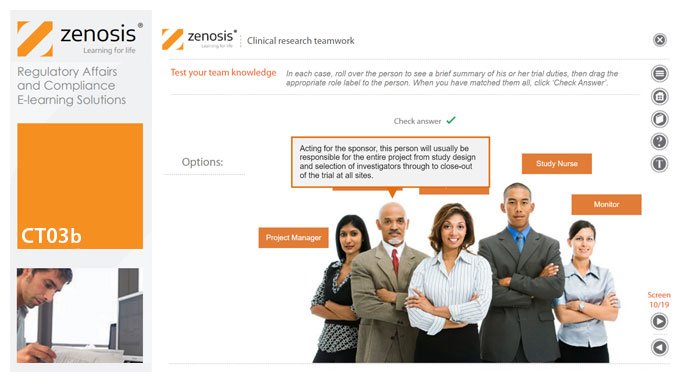
CT03b - Clinical research teamwork
By Zenosis
A clinical trial, particularly a late-phase commercial study, is a major project requiring collaboration between the sponsor and staff or contractor, on the one hand, and the clinical investigator(s) and other healthcare professionals on the other. Good communication among all parties is essential. In this short course we introduce the major roles in a typical clinical research project and outline their duties.
Driver Safety Awareness
By Imperial Academy
Level 3 QLS Endorsed Course | CPD & CiQ Accredited | Audio Visual Training | Free PDF Certificate | Lifetime Access

Dealing Conflict at Work, Stress Management and Communication for HR
By Imperial Academy
3 QLS Endorsed Diploma | QLS Hard Copy Certificate Included | 10 CPD Courses | Lifetime Access | 24/7 Tutor Support

Digital Marketer, Affiliate Marketing & Search Engine Optimisation
By Imperial Academy
Level 3, 4 & 7 QLS Endorsed Diploma | QLS Hard Copy Certificate Included | Plus 10 CPD Courses | Lifetime Access

QUALIFI Level 4 Diploma in Psychology
By School of Business and Technology London

QUALIFI Level 4 Diploma in Information Technology- Networking
By School of Business and Technology London

OTHM Level 4 Diploma in Project Management
By School of Business and Technology London

QUALIFI Level 5 Diploma in Information Technology- Networking
By School of Business and Technology London

QUALIFI Level 5 Diploma in Information Technology- Web Design
By School of Business and Technology London
