- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
1208 Courses delivered Online
Explore the fascinating world of archaeology with our hands-on training course in Fieldwork Techniques. From preliminary research to cutting-edge technology applications, dive into excavation, artefact analysis, conservation, and specialized site work. Gain practical skills in field recording and documentation, while incorporating modern technology like GIS and 3D modeling. Unearth a comprehensive understanding of archaeological methods for a fulfilling career or enrich your expertise. Join us for a transformative journey where history comes to life through experiential learning.

BCS Practitioner Certificate in Requirements Engineering
By Career Smarter

C10M02 - Actuation of door release mechanisms - BS 7273-4 (online)
By Ember Compliance
Learners will develop knowledge from the FD&FA foundation courses (C01 modules 1 and 2) and/or industry experience with specific regard to more complex system design considerations.

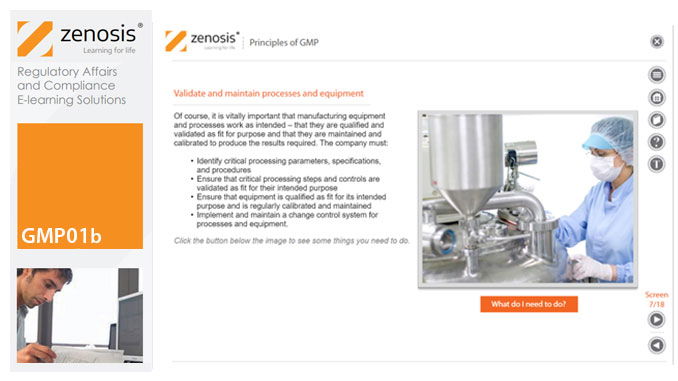
GMP01b - Principles of GMP
By Zenosis
In this short course we present an overview of the main principles of GMP, and we outline some things that manufacturing personnel need to do to comply with requirements. We identify the principal goals of GMP as: prevention of contamination; prevention of mix-ups; scrupulous documentation; validation and maintenance of processes and equipment; quality assurance by an independent unit; and training. We place GMP in the context of a company’s quality management system.
Data Entry Administration & Office Admin Assistant Course
By Training Express

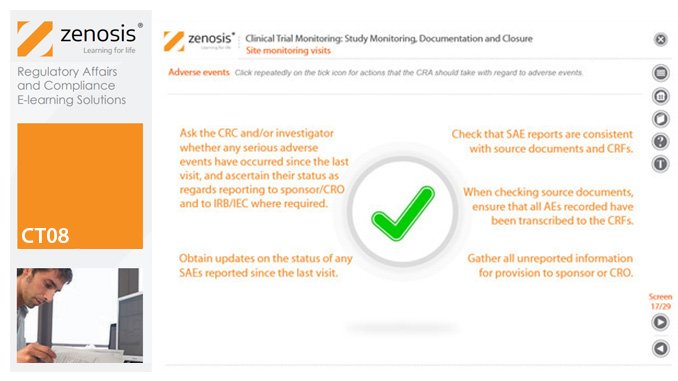
CT08: Clinical Trial Monitoring: Study Monitoring, Documentation and Closure
By Zenosis
The sponsor of a clinical trial must arrange for it to be monitored throughout its duration to ensure that the rights and wellbeing of subjects are protected, the trial data are accurate, complete and verified from source documents, and the conduct of the trial complies with the study protocol, Good Clinical Practice and regulatory requirements. In this module we describe how a Clinical Research Associate (CRA) monitors an ongoing trial to its conclusion.
Assuring Quality Through Acceptance Testing - Making Sure that the Business Problem is Solved: On-Demand
By IIL Europe Ltd

Level 4 Diploma in Import/Export
By Compliance Central

Introduction to Rust Programming
By Packt
This course will take you from the basics of Rust to understanding how to use all its features effectively for writing clear and effective code. With the help of engaging exercises and explanations, you'll learn the basic syntaxes, iterators, generics, borrow checker, code documentation, pointer lifetimes, and much more.

