- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
How to help young people who self harm
By Human Givens College

Achieving Outputs in a Complex World
By Mpi Learning - Professional Learning And Development Provider
Leadership is about 'getting stuff done'. This course concentrates on just that. In a complicated world, it helps leaders to ensure that the right stuff gets done at the right time and in the right way.

Learn Unity Game Development - Build Six Games with Unity 2023
By Packt
Get ready to dive into the exciting world of Unity game development and C# scripting! With a hands-on approach, you will craft a variety of thrilling 2D and 3D games using Unity and C#. Uncover the art of building and exporting games to the Android mobile platform. This course is tailor-made for someone who wants to learn Unity and C# through real-world projects.

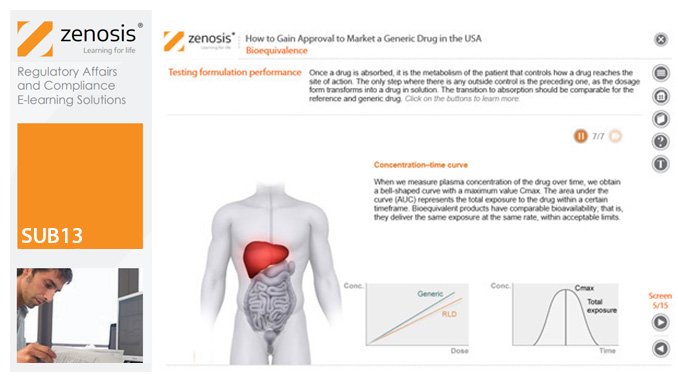
SUB13: How to Gain Approval to Market a Generic Drug in the USA
By Zenosis
This module outlines the legislative and regulatory context for the development of generic drugs and describes the essential role of the Abbreviated New Drug Application (ANDA) in gaining marketing approval. The use of information in the ‘Orange Book’ is explained, as is the role of patent certification in the application. The importance of establishing bioequivalence between a generic and its reference product is emphasised. The module specifies the content and format requirements for an ANDA submission and describes the FDA’s review and approval process. An outline is given of the Generic Drug User Fee Amendments (GDUFA) and the law’s effects on industry players.
Couples Therapy: Is Narcissism the problem?
By Human Givens College

Complete Maya Beginner to Advanced Training
By ATL Autocad Training London
Secrets of Acting Shakespeare: First Folio Technique
By Actors Studio