- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
327 Courses delivered Online
VAL01: Introduction to Validation
By Zenosis
Validation of equipment, services, systems and processes is vitally important in the medicines and healthcare products industries. Regulatory authorities require documented evidence that manufacturing processes will consistently result in products meeting predetermined quality standards. This module provides an introduction to validation and to the regulations and guidance that apply to it. It describes the activities of a typical validation team as they carry out a project for a pharmaceutical company.
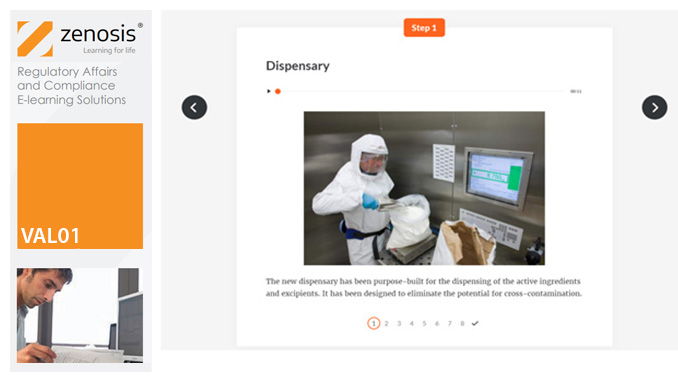
VAL02: Validation Plans and Documentation
By Zenosis
Essential to validation is the provision of documented evidence verifying that manufacturing processes will consistently result in products meeting predetermined quality standards. This module describes the purpose, content and use of validation master plans, project validation plans, and other documentation for validation projects in the medicines and healthcare products industries. It describes the activities of a typical validation team as they carry out a project for a pharmaceutical company.
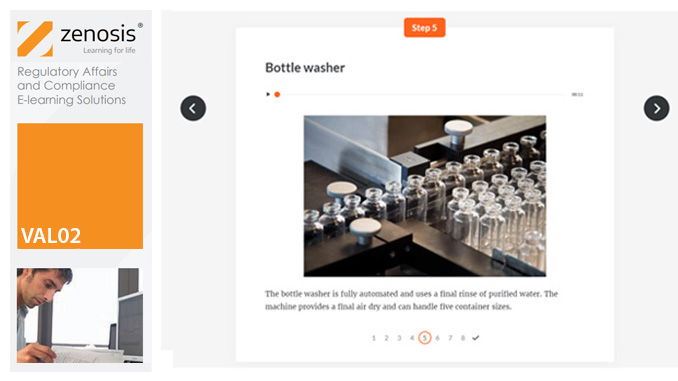
VAL05: Equipment Cleaning Validation
By Zenosis
Manufacturers of medicines and healthcare products must establish, validate and maintain an equipment cleaning programme. This is a regulatory requirement because validated cleaning procedures contribute to the assurance of product purity and safety. This module provides a comprehensive account of equipment cleaning validation requirements and procedures. It follows the work of a pharmaceutical company's validation team as they establish and validate the cleaning program for a new production line.
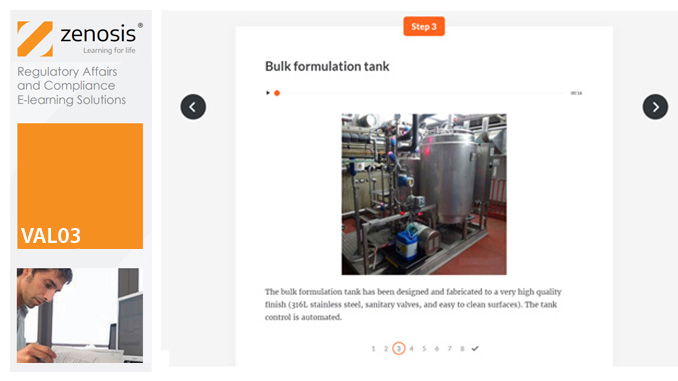
VAL03: Commissioning and Installation Qualification
By Zenosis
Before equipment can be used routinely in production, it must first be commissioned and, if necessary, undergo Installation Qualification (IQ). This module describes commissioning and IQ requirements and procedures in the medicines and healthcare products industries. It follows the activities of a typical validation team as they carry out a project for a pharmaceutical company.
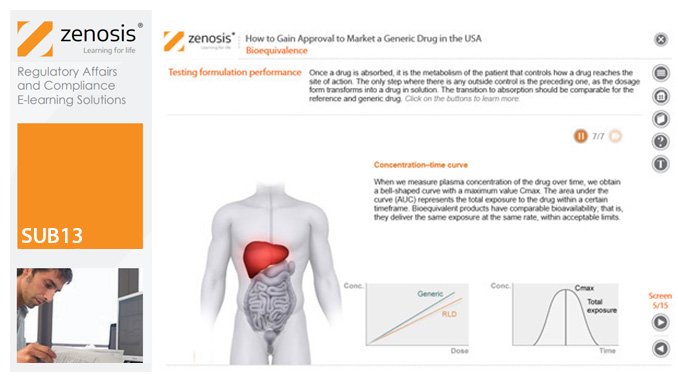
SUB13: How to Gain Approval to Market a Generic Drug in the USA
By Zenosis
This module outlines the legislative and regulatory context for the development of generic drugs and describes the essential role of the Abbreviated New Drug Application (ANDA) in gaining marketing approval. The use of information in the ‘Orange Book’ is explained, as is the role of patent certification in the application. The importance of establishing bioequivalence between a generic and its reference product is emphasised. The module specifies the content and format requirements for an ANDA submission and describes the FDA’s review and approval process. An outline is given of the Generic Drug User Fee Amendments (GDUFA) and the law’s effects on industry players.
VAL06: Computer Systems Validation, Part 1: Planning
By Zenosis
In the medicines and healthcare products industries, computerised systems used in automated manufacturing or laboratory processes to which Good Manufacturing Practice requirements apply need to be validated. This module describes the planning of such validation. It follows the work of a pharmaceutical company's team as they validate the dispensary control system for a new production line.

Medical Teaching Diploma - CPD Certified Course
By Training Express

UKMLA/PLAB 1 & 2 Combined Package
By Hive Medical Academy
Services Included PLAB1 Premium; ✔ 24/7 access to Course Tutors via WhatsApp. ✔ Live UKMLA/PLAB1 teaching sessions x 3. ✔ UKMLA/PLAB1 Complete Video Course. ✔ 10 x UKMLA/PLAB1 Mock Tests. ✔ 5K plus, the latest PLAB1/UKMLA MCQs. ✔ UKMLA/PLAB1 Complete Audio Course. ✔ 250 Digital Flashcards for rapid revision, (Clinical & Pharmacology) PLAB2 Premium; ✔ Intense 10-day face-to-face teaching programme. ✔ SimMan and Manikin teaching and practice. ✔ PLAB2 Online video course. ✔ 2 x PLAB2 Mock Exam sessions – booked when it suits you as per your exam date. ✔ 2 X UKMLA/PLAB2 Audio courses. ✔ More than 1,000 Revision Flashcards, ➢ Clinical ➢ Data Gathering, ➢ Cases & Explanation ➢ Examination & Procedures ➢ Differential Diagnosis ➢ NICE/CKS Guidelines ✔ PLAB2/UKMLA PRACTICE CASES. Key Points ✔ The most comprehensive all-in-one package for PLAB 1 Focus on both PLAB 1 knowledge and exam technique. ✔ Teaching by senior NHS clinicians including previous PLAB examiners ✔ Teaching based on UK guidelines, regularly updated Chapter-based video and audio teaching for ease of use. ✔ Multiple teaching formats for optimum preparation (live, video, audio, mocks, flashcards) ✔ Most comprehensive all-in-one package for PLAB2. ✔ Teaching by senior NHS clinicians including previous PLAB examiners. ✔ Focus on PLAB2 knowledge, technique and time management. ✔ High-quality SimMan and mannikins to learn and practice on. ✔ Non-scripted, principles-based approach. ✔ Practice-centric course – with role-play and feedback from day 1. ✔ Continuous role-plays, mock cases, practice and feedback of all key scenario types. ✔ Increase confidence in a full range of scenario types and situations. ✔ Cover areas that are often worried about eg prescribing, teaching emergency scenarios. ✔ Teaching based on UK guidelines, regularly updated. ✔ Chapter-based video and audio teaching for ease of use. ✔ Ongoing support after the academy concludes, up to exam day. ✔ All material is available for 12 months, with no limits to use at this time.


